Computational Modeling of the Effects of Inflammatory Response and Granulation Tissue Properties on Human Bone Fracture Healing
Bone healing process includes four phases: inflammatory response, soft callus formation, hard callus development, and remodeling. Mechanobiological models have been used to investigate the role of various mechanical and biological factors on the bone healing. However, the initial phase of healing, which includes the inflammatory response, the granulation tissue formation and the initial callus formation during the first few days post-fracture, are generally neglected in such studies. In this study, we developed a finite-element-based model to simulate different levels of diffusion coefficient for mesenchymal stem cell (MSC) migration, Young’s modulus of granulation tissue, callus thickness and interfragmentary gap size to understand the modulatory effects of these initial phase parameters on bone healing. The results showed that faster MSC migration, stiffer granulation tissue, thicker callus and smaller interfragmentary gap enhanced healing to some extent. After a certain threshold, a state of saturation was reached for MSC migration rate, granulation tissue stiffness and callus thickness. Therefore, a parametric study was performed to verify that the callus formed at the initial phase, in agreement with experimental observations, has an ideal range of geometry and material properties to have the most efficient healing time. Findings from this paper quantified the effects of the healing initial phase on healing outcome to better understand the biological and mechanobiological mechanisms and their utilization in the design and optimization of treatment strategies. Simulation outcomes also demonstrated that for fractures, where bone segments are in close proximity, callus development is not required. This finding is consistent with the concepts of primary and secondary bone healing.
💡 Research Summary
This paper presents a comprehensive finite‑element (FE)‑based mechanobiological model that explicitly incorporates the early inflammatory phase of bone fracture healing—an aspect often omitted in previous studies. Building on the Lacroix & Prendergast (2002) framework, the authors modeled a human long bone as a hollow cylinder cut transversely, discretized with 4‑node quadrilateral displacement and pore‑pressure elements. Five tissue types (cortical bone, marrow, granulation tissue, fibrous tissue, cartilage) were represented as linear poroelastic biphasic materials with properties taken from the literature (Table 1).
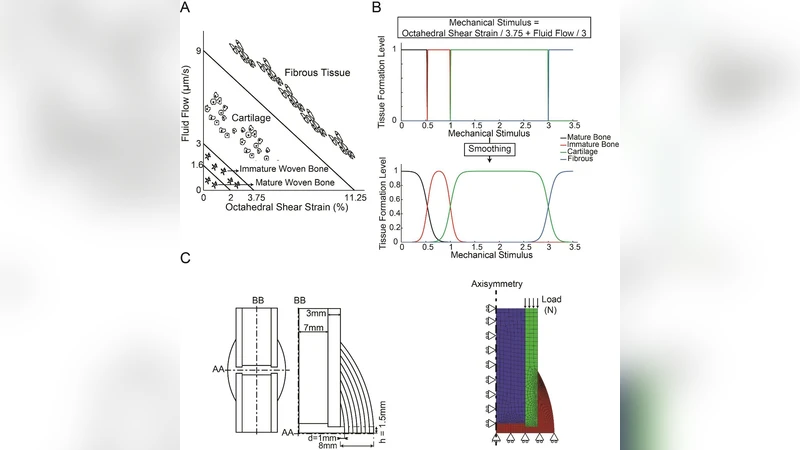
The mechanobiological regulation follows Prendergast et al. (1997), where high mechanical stimuli promote fibrous tissue, intermediate stimuli favor cartilage, and low stimuli induce bone formation. This rule was smoothed using the Sapotnick & Nackenhorst (2015) formulation to avoid abrupt transitions.
Four key early‑phase parameters were varied systematically: (1) MSC diffusion coefficient (D) ranging from 0.001 to 100 mm² day⁻¹, (2) Young’s modulus of granulation tissue (Eg) from 0.01 to 2 MPa, (3) initial callus thickness (d) from 1 to 8 mm, and (4) interfragmentary half‑gap (h) from 0.5 to 4 mm. For each combination, the model simulated 120 days of healing, updating MSC density via a separate diffusion analysis, then computing octahedral shear strain and fluid flow to drive tissue differentiation. Healing milestones were defined as the days when (i) a cartilaginous callus (CC) formed (elements >10 MPa connecting the fragments), (ii) bony bridging (BB) occurred (elements >2 GPa), and (iii) a complete bony callus (BC) filled the entire callus region (all elements >2 GPa).
Results showed clear, non‑linear dependencies. MSC diffusion coefficients between 0.1 and 1 mm² day⁻¹ yielded normal healing (CC ≈ 23–25 days, BB ≈ 46–48 days, BC ≈ 66–70 days). Below 0.01 mm² day⁻¹, cell density never reached the 50 % threshold, leading to delayed or absent bony callus even after 120 days. Increasing D beyond 10 mm² day⁻¹ produced no further acceleration, indicating a saturation effect.
Granulation tissue stiffness behaved similarly. Values ≤ 0.2 MPa produced negligible changes in healing times, whereas raising Eg from 0.2 to 2 MPa advanced CC by ~10 days, BB by ~16 days, and BC by ~24 days. Conversely, reducing Eg to 0.1 MPa dramatically increased interfragmentary strain and fluid flow, slowing differentiation.
Callus thickness proved the most influential parameter. A 1 mm callus resulted in a fibrous, non‑union outcome. A 2 mm callus achieved full bony union in about four months, while thicknesses of 3–6 mm accelerated the process to 6–10 weeks. Thicknesses of 7–8 mm further shortened the timeline to less than one month, but beyond 6 mm the benefit plateaued. Larger callus also reduced early strain and fluid flow, creating a more favorable mechanical environment.
Interfragmentary gap size dictated whether a callus was required at all. Gaps ≤ 1 mm (h ≤ 0.5 mm) allowed direct bone contact and primary healing without callus formation, consistent with classic primary healing concepts. Larger gaps necessitated callus formation, and the optimal callus geometry (3–6 mm thickness, appropriate stiffness) emerged as a key determinant of efficient secondary healing.
The authors interpret these findings as evidence that early‑phase biological and mechanical factors interact to set a “window of opportunity” for optimal healing. MSC migration, granulation tissue stiffness, and callus geometry each have threshold values beyond which additional increases confer no benefit—a saturation phenomenon. Moreover, the model predicts that an “ideal” initial callus exists, balancing sufficient mechanical support with minimal impediment to strain‑driven differentiation.
Clinically, the study suggests that interventions targeting the inflammatory phase (e.g., anti‑inflammatory drugs, growth‑factor delivery) must preserve adequate MSC migration and granulation tissue compliance to avoid delayed union. Surgical fixation strategies that minimize interfragmentary gaps can promote primary healing and reduce the need for excessive callus formation. The parametric framework could be employed to personalize treatment plans, testing the impact of pharmacologic or mechanical modifications on early healing trajectories.
Limitations include the use of linear elastic, isotropic material models, omission of angiogenesis, immune cell dynamics, and biochemical signaling pathways. Future work should integrate these biological processes, incorporate patient‑specific geometry and material heterogeneity, and validate predictions against longitudinal in‑vivo imaging data.
In summary, this paper quantitatively demonstrates how the early inflammatory response and granulation tissue properties shape the entire fracture healing cascade, providing a mechanistic basis for optimizing both biological and mechanical therapeutic strategies.
Comments & Academic Discussion
Loading comments...
Leave a Comment