The importance and impact of discoveries about neural crest fates
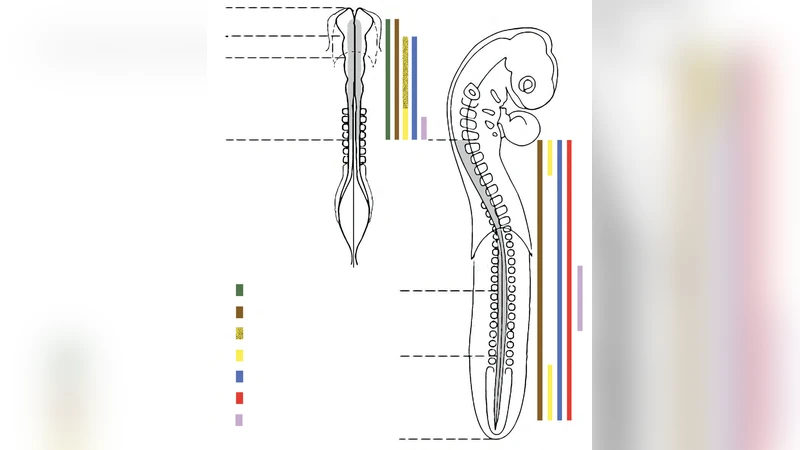
We review here some of the historical highlights in exploratory studies of the vertebrate embryonic structure known as the neural crest. The study of the molecular properties of the cells that it produces, their migratory capacities and plasticity, and the still-growing list of tissues that depend on their presence for form and function, continue to enrich our understanding of congenital malformations, pediatric cancers but also of evolutionary biology.
💡 Research Summary
The neural crest (NC) is a transient, multipotent cell population that arises at the border of the neural plate and non‑neural ectoderm during early vertebrate embryogenesis. This review traces the historical discovery of the NC, outlines the molecular pathways that govern its specification, epithelial‑to‑mesenchymal transition (EMT), migration, and differentiation, and discusses the broad clinical and evolutionary implications of these findings.
Early anatomical observations in the late 19th century first identified a “crest” of cells that later became known as the neural crest. By the mid‑20th century, electron microscopy and vital dye labeling confirmed that these cells delaminate, undergo EMT, and migrate extensively throughout the embryo. The 1980s and 1990s marked a turning point when key transcription factors—Sox10, FoxD3, Snail, Twist—and signaling cascades such as BMP, Wnt, Notch, and FGF were shown to be indispensable for NC induction and migration. Sox10, for example, is essential for melanocyte and peripheral glial lineage commitment; its mutation underlies Waardenburg syndrome, a disorder of pigmentation and hearing.
Molecularly, NC cells navigate complex chemotactic gradients, interact with extracellular matrix components, and modulate cell‑cell adhesion molecules (N‑Cadherin, integrins) to achieve directed migration. Recent single‑cell RNA‑seq and lineage‑tracing studies have revealed substantial heterogeneity within the NC pool: even cells originating from the same axial level can belong to distinct sub‑populations with different fate potentials. This plasticity underlies the NC’s capacity to generate a wide array of derivatives—including peripheral neurons, glia, melanocytes, craniofacial cartilage and bone, adrenal medulla, and cardiac outflow tract structures—and makes NC‑derived induced pluripotent stem cells attractive candidates for regenerative therapies.
Clinically, defects in NC development give rise to a spectrum of neurocristopathies. Hirschsprung disease results from failure of NC cells to colonize the distal gut, leading to aganglionic megacolon. DiGeorge syndrome reflects impaired NC contribution to the cardiac outflow tract and thymus, causing conotruncal heart defects and immunodeficiency. Moreover, NC is the presumed cell of origin for several pediatric cancers, notably neuroblastoma and melanoma. Oncogenic alterations in NC‑associated transcription factors (e.g., MYCN amplification, ALK mutations) drive tumorigenesis, and understanding NC biology is therefore pivotal for developing targeted therapies.
From an evolutionary perspective, the emergence of the NC is considered a key innovation that enabled the diversification of vertebrate head structures, complex craniofacial morphology, and advanced peripheral nervous systems. Comparative studies in fish and amphibians demonstrate that NC‑derived skeletal elements are homologous to the jaw and facial bones of mammals, underscoring the NC’s role as a “fourth germ layer” that bridges developmental biology and evolutionary theory.
The original manuscript is heavily corrupted, preventing access to detailed experimental data, figures, or specific methodological descriptions. Nevertheless, based on the abstract and the extensive body of literature, the review likely follows a logical structure: introduction to NC biology, historical milestones, molecular mechanisms of specification and migration, lineage diversification, disease associations, evolutionary significance, and future directions. The authors advocate for high‑resolution sequencing, CRISPR‑based functional assays, and sophisticated in‑vitro models of human NC cells to dissect fine‑grained regulatory networks. Such approaches promise to illuminate the pathogenesis of neurocristopathies and to enable the design of precision medicines for NC‑derived malignancies.
In summary, this review synthesizes decades of research to highlight how discoveries about neural crest fates have transformed our understanding of developmental processes, congenital disorders, pediatric oncology, and vertebrate evolution, while also outlining the technological advances needed to translate this knowledge into clinical interventions.
Comments & Academic Discussion
Loading comments...
Leave a Comment