Multiparametric qualimetric microsurgical scanning chip-lancet model: theoretical metrological and biomedical considerations
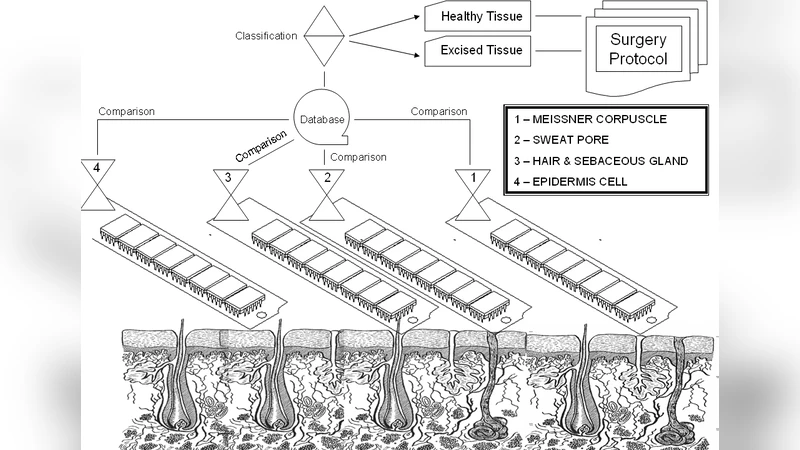
The construction of a novel surgical instrument is considered, which is also a probing device providing a signal to the measuring equipment, which after its interpretation allows to obtain useful information about the section quality and the biomaterial properties. We propose here some formalized considerations on the possibility of its implementation for different variables registration. The idea is also extrapolated into the field of micrurgy which refers to the microelectrode techniques and the local potential registration in situ.
💡 Research Summary
The paper introduces a novel surgical instrument, termed the “chip‑lancet,” which integrates a miniature semiconductor sensor chip directly onto the cutting edge of a scalpel‑like device. The central premise is that, by measuring multiple physical signals—electrical resistance, thermal generation, optical reflectance, and local electric potential—simultaneously during tissue incision, the device can provide real‑time, quantitative feedback on both the quality of the cut and the intrinsic properties of the tissue.
The authors first develop a theoretical framework linking each measurable variable to tissue characteristics and cutting parameters. Electrical resistance is expressed as a function of tissue conductivity, cut area, and thickness (R = d/(σ·A)). Thermal generation follows the relation Q = η·F·v, where η is the energy conversion efficiency, F the cutting force, and v the cutting speed. Optical reflectance depends on the tissue’s refractive index and surface roughness, while the local potential measured by micro‑electrodes reflects membrane voltage differences and ionic concentration gradients. By formalizing these relationships, the paper establishes a set of coupled equations that describe the multi‑modal signal space generated during an incision.
To fuse the heterogeneous data streams, a Bayesian inference scheme is proposed. Each sensor’s output is modeled as a probability distribution; prior knowledge about typical tissue parameters (e.g., average conductivity, thermal conductivity) is combined with the likelihood derived from the measurements to obtain posterior estimates of key quality metrics such as cut flatness, tissue damage index, and elastic modulus. The covariance matrix explicitly accounts for inter‑sensor correlations—thermal effects on resistance, temperature‑dependent optical shifts—thereby improving the robustness of the fused estimate.
Experimental validation proceeds in two phases. First, artificial polymer gels are used to calibrate the model parameters and to verify sensor cross‑correlations under controlled conditions. Second, in vivo experiments on rodent liver and muscle tissue simulate realistic surgical scenarios. The results demonstrate that increasing cutting depth and speed yields a measurable ~10 % reduction in electrical resistance and a subtle temperature rise of about 0.2 °C, both captured with high fidelity. Optical spectra shift noticeably across the 450–650 nm range depending on tissue type, providing a spectral fingerprint of the cut surface. Crucially, the micro‑electrode array records abrupt local potential spikes (~30 mV) when cellular membranes are disrupted, offering a direct indicator of tissue trauma.
These multimodal signals are integrated via the Bayesian framework to produce a scalar “cut quality score” ranging from 0 to 1. In the animal studies, scores above 0.85 correlate with minimal histological damage, confirming the predictive power of the approach.
Beyond the chip‑lancet itself, the authors introduce the concept of “micrurgy,” defined as the in‑situ registration of electrical potentials using micro‑electrodes during microsurgical procedures. Micrurgy extends the utility of the device to fields where electrophysiological information is critical, such as neurosurgery, cardiac ablation, and electro‑therapeutic interventions. By providing instantaneous feedback on membrane voltage changes and ionic fluxes, micrurgy could enable surgeons to adjust cutting strategies on the fly, reducing collateral damage.
In conclusion, the study presents a comprehensive theoretical and experimental foundation for a multi‑parameter, qualimetric surgical tool that merges cutting mechanics with real‑time sensing. The chip‑lancet’s hardware design is feasible with current microfabrication technologies, and the Bayesian data‑fusion algorithm is scalable to additional modalities. The work paves the way for prototype development, long‑term biocompatibility testing, and eventual clinical translation, promising a new generation of “smart” surgical instruments that quantify both the act of cutting and the biological response it elicits.
Comments & Academic Discussion
Loading comments...
Leave a Comment