Quantitative Phase Imaging and Artificial Intelligence: A Review
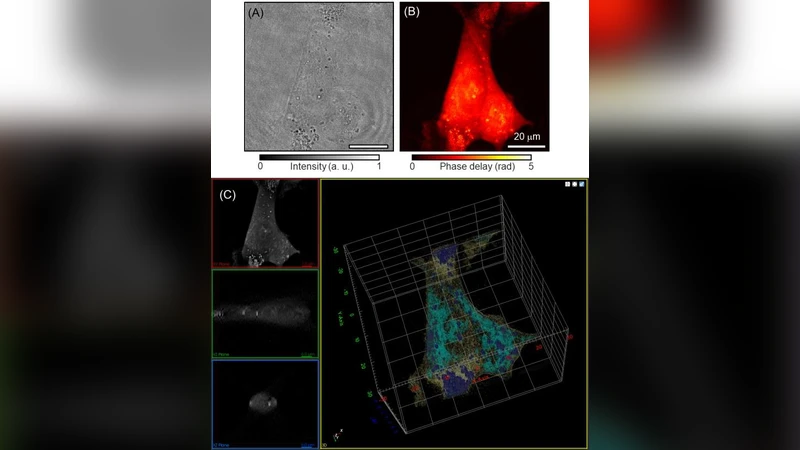
Recent advances in quantitative phase imaging (QPI) and artificial intelligence (AI) have opened up the possibility of an exciting frontier. The fast and label-free nature of QPI enables the rapid generation of large-scale and uniform-quality imaging data in two, three, and four dimensions. Subsequently, the AI-assisted interrogation of QPI data using data-driven machine learning techniques results in a variety of biomedical applications. Also, machine learning enhances QPI itself. Herein, we review the synergy between QPI and machine learning with a particular focus on deep learning. Further, we provide practical guidelines and perspectives for further development.
💡 Research Summary
The review provides a comprehensive overview of the rapidly evolving synergy between quantitative phase imaging (QPI) and artificial intelligence (AI), with a particular emphasis on deep learning. QPI is introduced as a label‑free, high‑throughput imaging modality capable of generating uniform, high‑quality data in two, three, and four dimensions. Its ability to capture the optical phase shift of transparent specimens translates into quantitative maps of refractive‑index distribution, which are intrinsically rich in biophysical information but traditionally require computationally intensive phase‑retrieval algorithms.
The authors then examine how modern machine‑learning techniques, especially convolutional neural networks (CNNs), generative adversarial networks (GANs), and vision transformers, have been adapted to process QPI data. Supervised approaches are highlighted for tasks such as cell type classification, disease diagnosis, and drug‑response screening, where large annotated datasets enable models to achieve accuracies surpassing conventional image‑analysis pipelines. Unsupervised and self‑supervised methods are discussed for feature extraction, clustering of phenotypic states, and dimensionality reduction, which are valuable when ground‑truth labels are scarce. The review also details the use of 3‑D CNNs and spatiotemporal architectures (e.g., CNN‑LSTM hybrids) for analyzing volumetric and time‑resolved QPI stacks, enabling real‑time monitoring of cellular dynamics, mitosis, and morphological changes under pharmacological perturbations.
A key insight is that AI does not merely consume QPI data; it actively enhances the imaging process itself. Hybrid physics‑driven deep networks incorporate known wave‑propagation constraints while learning data‑driven priors, resulting in faster, more robust phase‑retrieval, denoising, and super‑resolution. These models can operate under low‑light or low‑contrast conditions where traditional algorithms fail, thereby extending the practical applicability of QPI in live‑cell environments.
The review catalogs a broad spectrum of biomedical applications enabled by the QPI‑AI partnership. In pathology, AI‑augmented QPI discriminates malignant from benign tissue with high sensitivity, leveraging subtle refractive‑index variations invisible to intensity‑only microscopy. In hematology, label‑free QPI combined with deep classifiers identifies abnormal red‑blood‑cell morphologies associated with anemia and malaria. In drug discovery, high‑content QPI screens capture dynamic cellular responses, and AI models predict dose‑response curves, dramatically reducing assay time and reagent costs.
Practical guidelines are provided for researchers seeking to implement QPI‑AI pipelines. Recommendations cover data acquisition (standardized illumination, calibration, and augmentation), dataset curation (balanced class representation, metadata management), model selection (transfer learning from natural‑image pretrained weights versus training from scratch on QPI‑specific corpora), training strategies (cross‑validation, early stopping, hyperparameter optimization), and evaluation metrics (accuracy, ROC‑AUC, physical consistency checks). The authors stress the importance of model interpretability, suggesting saliency‑map techniques (Grad‑CAM, SHAP) to relate learned features back to biophysical phenomena, thereby fostering trust among clinicians.
Looking forward, the authors anticipate several trends that will shape the field. Multimodal integration—combining QPI with fluorescence, Raman, or electrical measurements—will provide complementary contrast mechanisms, while federated learning and privacy‑preserving techniques will enable collaborative model training across institutions without sharing raw patient data. Edge‑computing hardware and model compression will bring real‑time QPI analysis to point‑of‑care devices. Finally, the establishment of community‑wide standards for data formats, benchmark datasets, and regulatory guidelines is deemed essential for translating QPI‑AI technologies from the laboratory to clinical practice.
In summary, the paper argues that the convergence of quantitative phase imaging and artificial intelligence constitutes a transformative platform for label‑free, high‑content biomedical imaging. By jointly advancing imaging hardware, algorithmic intelligence, and application‑driven validation, the QPI‑AI ecosystem is poised to accelerate discovery, improve diagnostics, and enable new therapeutic screening paradigms.
Comments & Academic Discussion
Loading comments...
Leave a Comment