TrialChain: A Blockchain-Based Platform to Validate Data Integrity in Large, Biomedical Research Studies
The governance of data used for biomedical research and clinical trials is an important requirement for generating accurate results. To improve the visibility of data quality and analysis, we developed TrialChain, a blockchain-based platform that can be used to validate data integrity from large, biomedical research studies. We implemented a private blockchain using the MultiChain platform and integrated it with a data science platform deployed within a large research center. An administrative web application was built with Python to manage the platform, which was built with a microservice architecture using Docker. The TrialChain platform was integrated during data acquisition into our existing data science platform. Using NiFi, data were hashed and logged within the local blockchain infrastructure. To provide public validation, the local blockchain state was periodically synchronized to the public Ethereum network. The use of a combined private/public blockchain platform allows for both public validation of results while maintaining additional security and lower cost for blockchain transactions. Original data and modifications due to downstream analysis can be logged within TrialChain and data assets or results can be rapidly validated when needed using API calls to the platform. The TrialChain platform provides a data governance solution to audit the acquisition and analysis of biomedical research data. The platform provides cryptographic assurance of data authenticity and can also be used to document data analysis.
💡 Research Summary
The paper presents TrialChain, a blockchain‑based data‑governance platform designed to ensure the integrity and auditability of data generated in large‑scale biomedical research, specifically the China “Million Persons Project” (MPP) on cardiovascular disease. The authors integrate the platform into an existing data‑science environment (NDSP) that already includes a Hadoop distributed file system, Spark, Hive, and a high‑performance computing cluster for genomic and imaging analyses. TrialChain adopts a hybrid architecture that combines a private blockchain (MultiChain 1.0.2) with periodic anchoring to a public blockchain (Ethereum).
In the private layer, a master node creates the MultiChain network, defines chain parameters, and grants permissions to client nodes. Each functional component—REST API, administrative web portal, and data‑ingestion services—runs in its own Docker container and connects to a dedicated MultiChain client via RPC. This design provides fault tolerance (each node replicates the ledger), fine‑grained access control, and rapid, low‑cost transaction processing. All blockchain operations, including node management, are recorded immutably.
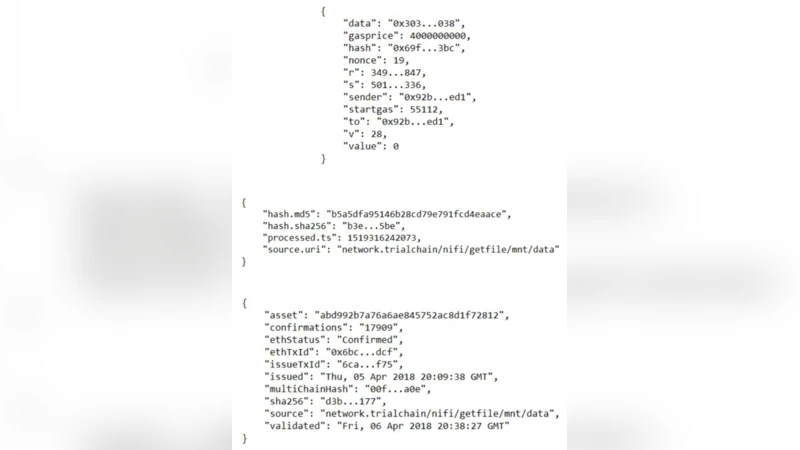
Data ingestion is handled by Apache NiFi. When a new file arrives in the NDSP, NiFi computes both an MD5 hash (used as the 32‑character Asset ID in MultiChain) and a SHA‑256 hash (stored as asset metadata for stronger cryptographic assurance). The hashes, together with a timestamp, source identifier, and optional metadata, are packaged into a JSON payload and sent via a POST request to a Falcon‑based REST API. The API registers the asset on the private MultiChain ledger, broadcasting the transaction to all peers.
To provide public verification, the platform periodically extracts the latest block hash (the “blockhash”) from the private chain and submits it as a transaction on the Ethereum mainnet. A Docker container runs a local Geth node; if the node is out‑of‑sync, the system can fall back to third‑party JSON‑RPC providers such as Infura or BlockCypher. A dedicated Ethereum wallet holds a small amount of Ether to pay the miner fee; the transaction contains the blockhash and optional metadata, is signed with the wallet’s private key, and is broadcast to the network. The transaction receipt, number of confirmations, and a link to etherscan.io are stored in a PostgreSQL database for later retrieval.
The administrative web portal (Flask‑based) allows users to query the status of any data asset by entering its MD5 hash. The portal looks up the asset on the private MultiChain node, determines whether the block containing the asset has been anchored to Ethereum, and returns the verification details (timestamp, transaction hash, confirmations, etherscan link). An embedded MultiChain Explorer provides direct visibility into the private ledger. The same verification functionality is exposed through a Python command‑line client, enabling automated scripts to validate assets programmatically.
Technical choices emphasize security, scalability, and cost‑effectiveness. Docker Compose orchestrates all services, ensuring reproducible deployment across Ubuntu 16.04 containers. The system leverages Kerberos for Hadoop authentication, SLURM for HPC job scheduling, and standard open‑source libraries (Web3.py, Savoir, Gevent) for blockchain interaction. By storing only cryptographic hashes on‑chain, the platform avoids the storage overhead of raw biomedical data while still providing immutable proof of existence and integrity.
In the discussion, the authors compare the private‑only versus hybrid approaches. Private blockchains offer low transaction fees and controllable access, but lack the universal immutability of public ledgers. Anchoring to Ethereum mitigates this limitation, delivering a publicly verifiable audit trail without exposing sensitive data. The paper also notes that the current implementation does not enforce data provenance beyond hash registration; future work may incorporate smart contracts for automated access control, standardized metadata schemas, and cross‑institutional federation to build a broader, interoperable data‑governance ecosystem.
Overall, TrialChain demonstrates a practical, production‑grade solution that integrates blockchain technology into existing biomedical research pipelines, delivering cryptographic assurance of data authenticity, transparent audit trails, and the ability for external stakeholders to independently verify that data have not been altered after collection. This contributes to higher trust in large‑scale clinical research outcomes and offers a template for similar initiatives in other domains.
Comments & Academic Discussion
Loading comments...
Leave a Comment