Micromechanical Experimental and Numerical Studies of Collagen Fibers Failure in Arterial Tissue
Arterial tissue failures lead to a number of clinical conditions that develop rapidly and unpredictably in vivo. Structural components and their interfacial mechanical strength of arterial tissue play a critical role in the process of arterail delamination. Therefore, there is a pressing need to understand the micromechanical mechanisms of arterial delamination. The objective of this study was to investigate various failure mechanisms (e.g. failure of collagen fibers) responsible for arterial interfacial delamination. In-situ tensile tests of fibers were performed on a micro-tester in the scanning electron microscope. A 3D unit cell model containing an individual fiber bridging two arterial tissue layers was constructed. An exponential cohesive zone model (CZM) was used to assess the stiffening and softening mechanical behaviors of collagen fiber bundles between the two arterial layers. An anisotropic constitutive model was implemented for characterizing the mechanical properties of the amorphous matrix which includes the fibrous cap and the underlying plaque tissue and a nonlinear elastic model was adopted for characterizing the mechanical properties of the fibers. The CZM and elastic parameter values of fibers were identified through an inverse boundary value approach that matches the load-displacement curves from simulation predictions of tensile test of collagen fibers with experimental measurements. The identified parameter values were then used as input in the 3D unit cell model, through which micromechanical factors affecting the resultant traction-separation relation for the unit cell were investigated via a parametric analysis. Results of the parametric analysis showed the applicability of the 3D unit cell model approach for evaluating the micromechanical mechanisms of arterial tissue failure processes.
💡 Research Summary
This paper addresses the critical need to understand the micromechanical mechanisms underlying arterial delamination, focusing specifically on the failure of collagen fibers that bridge arterial layers. The authors combined in‑situ tensile testing of collagen fiber bundles harvested from porcine abdominal aorta with a three‑dimensional finite‑element unit‑cell model that incorporates an exponential cohesive zone model (CZM) to capture the stiffening and softening behavior of fiber bundles at the interface between two arterial layers.
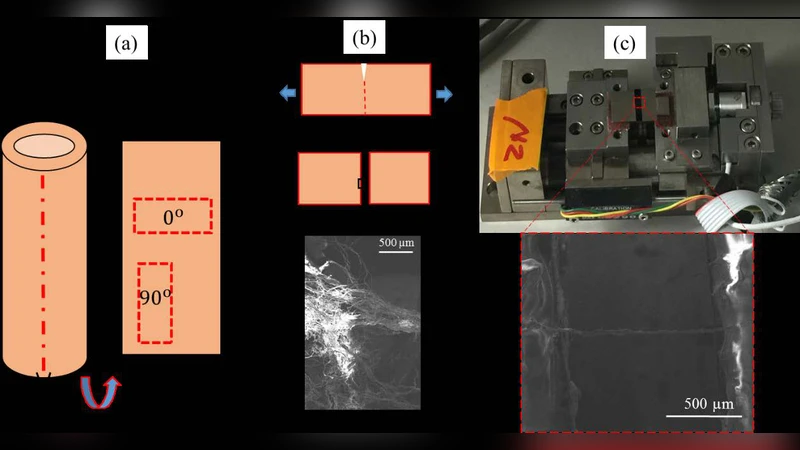
Experimental methods: Six specimens of collagen fiber bundles were isolated by tearing a 20 mm × 10 mm aortic strip and exposing the fibers on a freshly cut surface. The bundles were clamped in a Gatan micro‑tester (2 N load cell) placed inside a FEI Quanta 400 scanning electron microscope. Uniaxial tensile tests were performed at a constant displacement rate of 1 mm/min while recording load‑displacement data and high‑resolution video of deformation and fracture. Measured properties included fiber diameter (≈10–40 µm), failure strain (≈8–32 %), and ultimate tensile strength (≈10–212 MPa).
Constitutive modeling: The amorphous matrix (fibrous cap and underlying plaque) was described using the Holzapfel‑Gasser‑Ogden (HGO) anisotropic hyperelastic model, which accounts for two families of collagen fibers with dispersion parameter κ. Individual collagen fibers were modeled as nonlinear elastic rods with a Poisson’s ratio of 0.3. The interface between adjacent fibers was represented by an exponential CZM defined by cohesive strength σ_c, characteristic separation δ_c, and a damage variable d that evolves from 0 (undamaged) to 1 (fully separated). The CZM formulation follows the energy‑based expression ψ = σ_c δ_c exp(−δ/δ_c) and was implemented via a user‑element subroutine in ABAQUS.
Parameter identification: An inverse boundary‑value approach was used to calibrate the fiber elastic modulus and CZM parameters. The calibration minimized the root‑mean‑square error between simulated and experimental load‑point‑displacement curves, achieving an error below 0.5 %. Identified values were, for example, fiber modulus ≈150 MPa, σ_c ≈2 MPa, δ_c ≈0.02 mm, and η (weighting of normal vs. shear separation) set to 1.0.
Finite‑element unit‑cell analysis: A 3‑D unit cell containing a single collagen fiber bridging two arterial layers was constructed. The cell geometry matched the experimentally measured fiber dimensions and matrix thickness. Boundary conditions fixed the left end of the fiber and applied a prescribed displacement rate at the right end, replicating the tensile test. A parametric study varied (i) fiber diameter, (ii) inter‑fiber cohesive strength, (iii) matrix stiffness, and (iv) the CZM damage parameter η. The resulting traction‑separation curves were examined for initial stiffness, peak traction, and energy release rate. Key findings include: larger fiber diameters increase interfacial shear strength and initial stiffness; lower σ_c reduces stiffness but raises fracture energy, indicating a more ductile failure mode; a stiffer matrix amplifies overall cell stiffness and localizes deformation; higher η yields a more gradual damage evolution and broader strain distribution.
Implications: The results demonstrate that collagen fiber rupture and the strength of the fiber‑to‑fiber interface critically control the initiation and propagation of arterial delamination. The unit‑cell framework provides a bridge between microscale fiber mechanics and macroscale cohesive zone models used in plaque rupture simulations, offering a quantitative tool to predict how changes in fiber properties (e.g., due to aging, disease, or pharmacological treatment) affect arterial integrity.
Limitations and future work: The study examined only single‑fiber bundles, neglecting the complex network of intersecting fibers present in vivo. Tests were conducted under dry, room‑temperature conditions, omitting physiological blood pressure and flow effects. The CZM parameters were assumed rate‑independent, whereas real tissue exhibits viscoelastic and rate‑sensitive behavior. Future research should incorporate multi‑fiber network models, rate‑dependent constitutive laws, and fluid‑structure interaction to capture the full physiological environment.
In summary, this work provides a rigorous experimental‑numerical methodology to quantify collagen fiber failure and interfacial adhesion in arterial tissue, elucidates the micromechanical contributors to arterial delamination, and establishes a versatile modeling platform for investigating therapeutic interventions aimed at strengthening arterial walls.
Comments & Academic Discussion
Loading comments...
Leave a Comment