An inverse analysis of cohesive zone model parameter values for human fibrous cap mode I tearing
Atherosclerotic plaque failure results from various pathophysiological events, with the existence of fibrous cap mode I tearing in the arterial wall, having the potential to block the aortic lumen and correspondingly induce serious clinical conditions.The aim of this study was to quantify the interfacial strength and critical energy release rate of the fibrous tissue across the thickness. in this study, an inverse analysis method via finite element modeling and simulation approach was presented. A cohesive zone model (CZM) was applied to simulate the tearing of the fibrous cap tissue under uniaxial tensile tests along the circumferential direction. A fiber-reinforced hyperelastic model (Holzapfel-Gasser-Ogden) was implemented for characterizing the mechanical properties of bulk material. With the material parameter values of HGO model from inverse analysis process as the input for the bulk material, the interfacial strength and critical energy release rate along the tearing path or failure zones are obtained through the same method as material identification process of HGO model. Results of this study demonstrate the fibrous cap tissue tearing failure processes.
💡 Research Summary
This paper presents a comprehensive inverse analysis framework to quantify the interfacial strength and critical energy release rate (Gc) of human carotid artery fibrous caps undergoing Mode I tearing. The authors combine a bulk anisotropic hyperelastic model—the Holzapfel‑Gasser‑Ogden (HGO) formulation—with a cohesive zone model (CZM) that captures the progressive damage of the tearing interface.
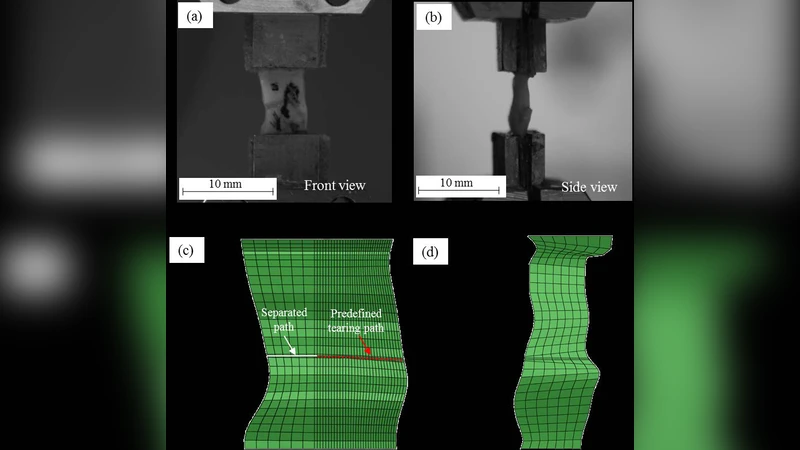
Experimental work involved five fresh human carotid specimens obtained during endarterectomy. Each fibrous cap was cut into circumferential strips, pre‑conditioned with five quasi‑static loading cycles (0.05 mm/s), and then a pre‑cut notch was introduced at the mid‑line to define the tearing path. Uniaxial tensile tests were performed at a constant displacement rate of 0.05 mm/s while recording load–displacement data and capturing front‑ and side‑view video.
Finite‑element models were built in ABAQUS (v6.13). The bulk tissue was discretized with eight‑node brick elements (C3D8H) and described by the HGO strain‑energy function, which incorporates a volumetric term (bulk modulus k) and a fiber‑reinforced term characterized by parameters k1, k2, fiber angle γ, and dispersion κ. The tearing interface was modeled using zero‑thickness eight‑node user‑defined elements (UEL) implementing an irreversible exponential CZM. The CZM law defines an effective traction σ as a function of effective separation δ, with two key parameters: the cohesive strength σc (interfacial strength) and the critical energy release rate Gc. A penalty stiffness K and a shape parameter β were fixed (K = 10 N/mm³, β = 1) to simplify the inverse problem.
The inverse analysis proceeded in two stages. First, the HGO parameters were identified by fitting simulated load‑point displacement curves to the experimental uniaxial tensile data. The optimization employed a MATLAB‑based Levenberg‑Marquardt algorithm that minimized the squared difference between simulated and measured forces. The resulting HGO parameters (Table 1) fell within ranges reported in the literature (k ≈ 2–5 kPa, k1 ≈ 300–3000 kPa, k2 ≈ 15–57, γ ≈ 57°–74°, κ ≈ 0.23–0.29).
Second, with the bulk parameters fixed, the CZM parameters σc and Gc were varied to reproduce the tearing test curves. The same optimization scheme was applied, now targeting the load‑displacement response of the tearing experiment. The identified interfacial strength was approximately 0.18–0.22 MPa, and the critical energy release rate was 0.20–0.26 N/mm (Table 2). Simulated tearing curves matched the experimental data with high correlation (R² > 0.95), and the model accurately captured the progression of the crack front as observed in the video recordings.
Key insights from the study include:
- Feasibility of inverse FE‑CZM identification – The combined HGO‑CZM framework can extract interface properties that are otherwise difficult to measure directly, providing essential inputs for patient‑specific plaque rupture simulations.
- Consistency with prior experimental observations – The obtained σc and Gc values align with those reported from direct delamination tests and from literature on arterial tissue fracture, supporting the validity of the methodology.
- Automation and reproducibility – By integrating ABAQUS with MATLAB for automated parameter sweeps and convergence checks, the authors establish a workflow that can be extended to larger datasets or other vascular tissues.
Limitations acknowledged by the authors are: the small sample size (n = 5) limiting statistical power; the assumption of a planar, straight tearing path, which neglects possible three‑dimensional crack branching; and the use of fixed β and K values, which may oversimplify the true variability of cohesive stiffness across the cap thickness. Future work should incorporate a larger cohort, explore variable penalty stiffness, and consider fully three‑dimensional crack propagation to enhance model fidelity.
In conclusion, the paper demonstrates that an inverse analysis coupling a fiber‑reinforced hyperelastic bulk model with a cohesive zone interface can reliably quantify the mechanical parameters governing fibrous cap tearing. These parameters are crucial for predictive modeling of plaque rupture, informing stent design, and potentially guiding personalized therapeutic strategies for atherosclerotic disease.
Comments & Academic Discussion
Loading comments...
Leave a Comment