Angiogenic Factors produced by Hypoxic Cells are a leading driver of Anastomoses in Sprouting Angiogenesis---a computational study
Angiogenesis - the growth of new blood vessels from a pre-existing vasculature - is key in both physiological processes and on several pathological scenarios such as cancer progression or diabetic retinopathy. For the new vascular networks to be functional, it is required that the growing sprouts merge either with an existing functional mature vessel or with another growing sprout. This process is called anastomosis. We present a systematic 2D and 3D computational study of vessel growth in a tissue to address the capability of angiogenic factor gradients to drive anastomosis formation. We consider that these growth factors are produced only by tissue cells in hypoxia, i.e. until nearby vessels merge and become capable of carrying blood and irrigating their vicinity. We demonstrate that this increased production of angiogenic factors by hypoxic cells is able to promote vessel anastomoses events in both 2D and 3D. The simulations also verify that the morphology of these networks has an increased resilience toward variations in the endothelial cell’s proliferation and chemotactic response. The distribution of tissue cell`s and the concentration of the growth factors they produce are the major factors in determining the final morphology of the network.
💡 Research Summary
This study investigates how angiogenic factors released by hypoxic tissue cells drive the formation of anastomoses—connections between growing vascular sprouts—during sprouting angiogenesis. Using a comprehensive computational framework that extends a phase‑field model, the authors represent the vascular network with an order parameter φ and the distribution of tissue cells with a second order parameter ψ. φ = +1 denotes vessel interior, –1 the surrounding matrix; ψ = +1 marks hypoxic cells, –1 the rest of the domain. The model couples these fields with a diffusion equation for an angiogenic factor (principally VEGF), which is produced at a constant rate T_s by any hypoxic cell that has not yet been irrigated by functional blood flow. When a newly formed vessel reaches a hypoxic region and supplies oxygen, the source term for that region is switched off, thereby reproducing the biological feedback that VEGF production ceases once tissue is perfused.
Endothelial cell (EC) proliferation is modeled as a function α(T) of the local VEGF concentration, rising with T until a saturation threshold T_p is reached. Tip cells are generated only when three conditions are satisfied: (i) VEGF concentration exceeds a minimal value T_c, (ii) the local VEGF gradient exceeds a minimal magnitude G_m, and (iii) the candidate tip is at least 4 R_c away from any existing tip, ensuring a “salt‑and‑pepper” distribution of tip versus stalk cells. Tip cells migrate chemotactically along the VEGF gradient; their speed is proportional to the gradient up to a maximal value χ, after which it saturates. Physical interaction with tissue cells is also incorporated: a tip cannot penetrate a tissue cell and will glide around it if it comes within 2 R_c.
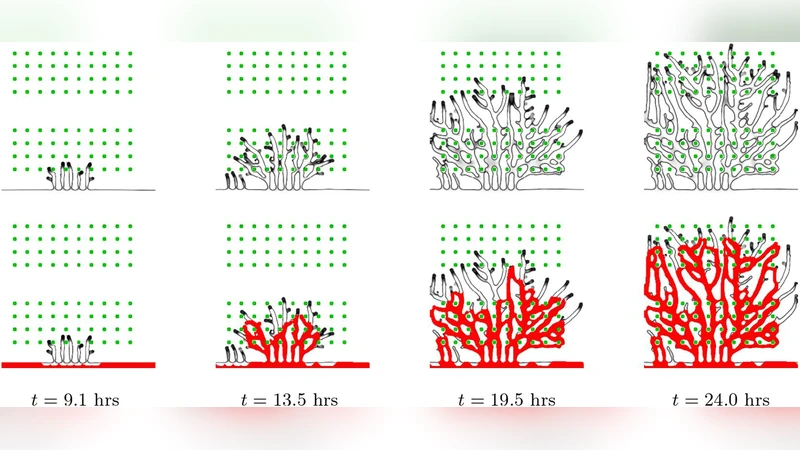
The authors performed simulations in both two‑dimensional (2‑D) and three‑dimensional (3‑D) domains. Hypoxic cells were placed either randomly or on regular lattices, while the vascular system was initialized with a single inlet at the top of the domain and a single outlet at the bottom. Blood flow was estimated post‑hoc by extracting the medial lines of the vessel network, identifying bifurcations, and assigning flow rates based on hydraulic resistance of each segment. Only vessels that belong to a closed loop (i.e., have flow) are considered perfused; these loops suppress VEGF production in the cells they irrigate, creating a negative feedback loop that limits further sprouting.
Key findings include:
-
VEGF‑driven anastomosis – When hypoxic cells continuously secrete VEGF until perfusion, the resulting concentration gradients guide tip cells toward one another, dramatically increasing the frequency of sprout‑sprout contacts and subsequent anastomoses. This effect is observed in both 2‑D and 3‑D simulations, but is especially pronounced in 3‑D where the spatial distribution of hypoxic sources creates a network of intersecting gradients that naturally steer sprouts into a lattice‑like meshwork.
-
Robustness to cellular parameters – The authors systematically varied endothelial proliferation rates (α_p) and chemotactic sensitivity (χ). In the presence of sustained VEGF production, the final vascular topology remained largely unchanged across a wide parameter range, indicating that the chemical gradient supersedes the mechanical and proliferative cues in determining network morphology. In contrast, when VEGF production was halted as soon as a sprout reached a hypoxic cell, the network became highly sensitive to these parameters, often failing to form closed loops.
-
Feedback between flow and VEGF – Once a loop is established, blood flow through that loop reduces hypoxia in the enclosed region, turning off VEGF sources. Consequently, the local VEGF gradient diminishes, curtailing the generation of new tip cells nearby. This self‑regulating mechanism prevents runaway angiogenesis and yields an efficient irrigated volume with minimal excess vasculature.
-
Implications for pathology and therapy – The model mirrors observations in tumor angiogenesis and diabetic retinopathy, where hypoxic tissue continuously secretes VEGF, leading to rapid formation of perfused vascular loops that sustain disease progression. Conversely, therapeutic strategies that blunt VEGF production or disrupt its gradient could impede anastomosis formation, offering a mechanistic rationale for anti‑VEGF treatments.
The paper also discusses limitations. Hypoxic cells are treated as immobile; in vivo they may migrate or remodel the extracellular matrix, potentially altering gradient fields. The Delta‑Notch/Jagged signaling that governs tip‑stalk selection is simplified to a distance‑based rule, whereas real endothelial cells exhibit complex lateral inhibition dynamics. Blood flow is modeled using a quasi‑steady resistance network, ignoring pulsatile pressure, shear stress, and vessel remodeling that occur over longer timescales.
In summary, this work provides a quantitative demonstration that sustained production of angiogenic factors by hypoxic cells is a primary driver of anastomosis formation during sprouting angiogenesis. By integrating chemical, mechanical, and hemodynamic feedbacks within a unified phase‑field framework, the authors show that VEGF gradients can dictate vascular network topology, confer robustness against variations in endothelial behavior, and shape the efficiency of tissue perfusion. These insights advance our understanding of vascular development and have direct relevance for designing anti‑angiogenic therapies in cancer, ocular disease, and other conditions characterized by pathological neovascularization.
Comments & Academic Discussion
Loading comments...
Leave a Comment