The application of collagen in advanced wound dressings
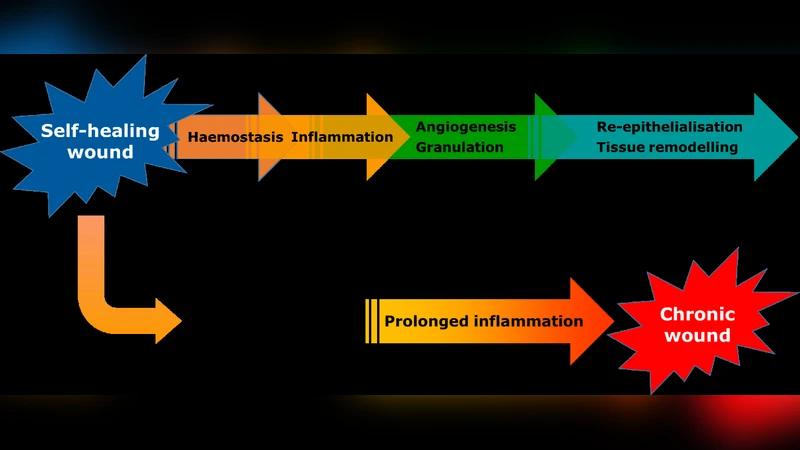
Chronic wounds fail to proceed through an orderly and timely self healing process, resulting in cutaneous damage with full thickness in depth and leading to a major healthcare and economic burden worldwide. In the UK alone, 200,000 patients suffer from a chronic wound, whilst the global advanced wound care market is expected to reach nearly $11 million in 2022. Despite extensive research efforts so far, clinically-approved chronic wound therapies are still time-consuming, economically unaffordable and present restricted customisation. In this chapter, the role of collagen in the extracellular matrix of biological tissues and wound healing will be discussed, together with its use as building block for the manufacture of advanced wound dressings. Commercially-available collagen dressings and respective clinical performance will be presented, followed by an overview on the latest research advances in the context of multifunctional collagen systems for advanced wound care.
💡 Research Summary
Chronic wounds—defined as lesions that fail to close within three months—represent a major clinical and economic challenge worldwide, affecting hundreds of thousands of patients in the UK alone and millions in the United States. Their pathophysiology is characterized by persistent inflammation, excessive matrix metalloproteinase (MMP) and reactive oxygen species (ROS) activity, and a deficit of growth factors, all of which impede the normal, overlapping phases of hemostasis, inflammation, proliferation, and remodeling. Conventional therapies such as negative‑pressure wound therapy, biologics, and standard dressings are limited by high cost, lengthy treatment times, and a lack of patient‑specific customization.
Collagen, the most abundant structural protein in the human body, accounts for roughly one‑third of total protein mass and two‑thirds of skin dry weight. Type I collagen, in particular, is the principal component of skin, tendon, and bone and plays a pivotal role in wound healing by providing a scaffold for fibroblast and keratinocyte migration, supporting angiogenesis through integrin‑binding motifs (RGD, GFOGER), and sequestering bioactive molecules. Its triple‑helical (Gly‑X‑Y) architecture, stabilized by covalent lysyl‑oxidase cross‑links, confers high mechanical strength and resistance to proteolysis, making it an ideal matrix for advanced dressings.
When used as a dressing material, collagen’s intrinsic hydrophilicity enables rapid absorption of wound exudate while maintaining a moist environment conducive to cell proliferation. Moreover, collagen can physically bind excess MMPs and growth factors, thereby reducing proteolytic degradation of newly formed tissue and prolonging the bioavailability of endogenous healing signals. This dual function—exudate management and biochemical modulation—distinguishes collagen dressings from traditional dry bandages.
Extraction of collagen for biomedical use typically employs mild acidic or enzymatic (collagenase) processes to preserve the triple‑helix. Harsh conditions (high temperature, extreme pH) cause denaturation to gelatin, which, although more soluble, exhibits lower mechanical integrity and faster in‑vivo degradation. Re‑assembly of extracted triple helices at physiological pH (7.4) and temperature (37 °C) can restore fibrillar structures, yet swelling and mechanical stability remain challenges that must be addressed through controlled cross‑linking and formulation strategies.
Source selection is critical. Bovine, porcine, and ovine collagens are widely used (e.g., in commercial products such as Promogran®, Biopad®, BiaStep®) but raise concerns about religious acceptability, immunogenicity, and transmissible spongiform encephalopathies. Alternative sources—fish, poultry, recombinant human collagen, and synthetic collagen‑mimetic peptides—offer reduced antigenicity and regulatory advantages, though they differ in amino‑acid composition, thermal stability, and mechanical properties. Genetic engineering now enables production of human‑type I collagen in plant or microbial hosts, providing a pathogen‑free, batch‑consistent raw material.
Recent research has moved toward multifunctional collagen dressings that actively remodel the wound microenvironment. Key innovations include:
- MMP‑responsive cross‑links: Peptide linkers cleavable by MMP‑9 allow selective degradation of the dressing at the wound site, triggering controlled release of embedded therapeutics.
- ROS‑scavenging components: Incorporation of catalytic nanoparticles (e.g., MnO₂) or polyphenolic antioxidants neutralizes excess ROS, protecting cells from oxidative damage.
- pH‑sensing colorimetric indicators: Embedded pH‑responsive dyes provide visual feedback on infection or inflammation status.
- Sustained delivery of growth factors and antimicrobial peptides: Electrostatic or covalent attachment of EGF, PDGF, LL‑37, etc., enables prolonged, localized bioactivity while protecting these labile molecules from rapid degradation.
Clinical data on existing collagen dressings demonstrate a 20–30 % improvement in healing rates and a reduction of treatment duration by approximately two weeks compared with standard care. Nevertheless, barriers remain: high manufacturing costs, variability in raw‑material quality, and the need for standardized regulatory pathways.
Future directions highlighted by the authors include (i) development of scalable, low‑cost extraction and purification pipelines that retain triple‑helical integrity; (ii) integration of advanced textile technologies such as 3D‑printing and electrospinning to fabricate dressings with tunable porosity, mechanical strength, and bioactive loading; and (iii) large‑scale, randomized controlled trials to rigorously assess safety, efficacy, and cost‑effectiveness across diverse patient populations. By addressing these challenges, collagen‑based advanced dressings have the potential to become a cornerstone of personalized chronic wound management, delivering both structural support and biochemical regulation in a single, patient‑friendly platform.
Comments & Academic Discussion
Loading comments...
Leave a Comment