Micro-sized cold atmospheric plasma source for brain and breast cancer treatment
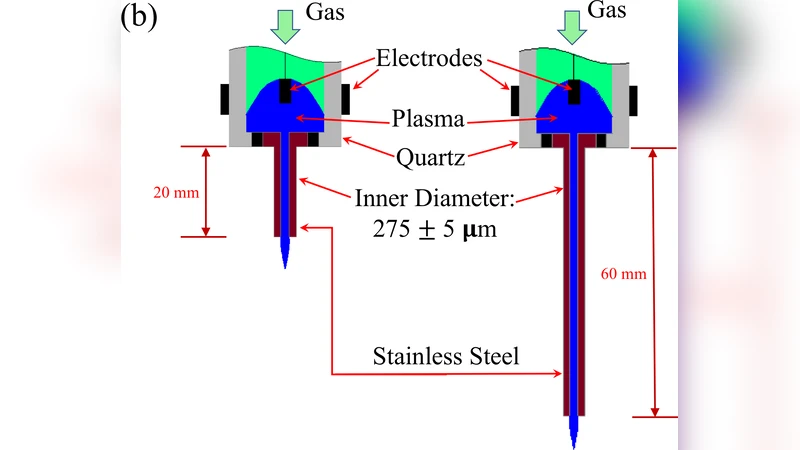
Micro-sized cold atmospheric plasma (uCAP) has been developed to expand the applications of CAP in cancer therapy. In this paper, uCAP devices with different nozzle lengths were applied to investigate effects on both brain (glioblastoma U87) and breast (MDA-MB-231) cancer cells. Various diagnostic techniques were employed to evaluate the parameters of uCAP devices with different lengths such as potential distribution, electron density, and optical emission spectroscopy. The generation of short- and long-lived species (such as hydroxyl radical (.OH), superoxide (O2-), hydrogen peroxide (H2O2), nitrite (NO2-), et al) were studied. These data revealed that uCAP treatment with a 20 mm length tube has a stronger effect than that of the 60 mm tube due to the synergetic effects of reactive species and free radicals. Reactive species generated by uCAP enhanced tumor cell death in a dose-dependent fashion and was not specific with regards to tumor cell type.
💡 Research Summary
This study introduces a micro‑sized cold atmospheric plasma (µCAP) device designed to broaden the therapeutic scope of cold atmospheric plasma (CAP) in oncology. Two µCAP configurations differing only in nozzle length—20 mm and 60 mm—were fabricated and characterized using electrical potential mapping, electron density measurements, and optical emission spectroscopy (OES). The shorter nozzle generated a markedly higher electric field (≈1.8‑fold) and electron density (3.2 × 10¹⁴ cm⁻³ versus 1.7 × 10¹⁴ cm⁻³ for the longer nozzle), leading to stronger emission of reactive species such as OH (309 nm) and atomic oxygen (777 nm). Chemical assays quantified short‑lived radicals (.OH, O₂⁻) and long‑lived species (H₂O₂, NO₂⁻) in the plasma‑treated media; the 20 mm device produced 2.3‑fold more .OH, 1.9‑fold more O₂⁻, 1.7‑fold more H₂O₂, and 1.5‑fold more NO₂⁻ than the 60 mm counterpart.
Biological efficacy was evaluated on human glioblastoma (U87) and triple‑negative breast cancer (MDA‑MB‑231) cell lines. Cells were exposed to µCAP for 30, 60, or 120 seconds, and viability was assessed by MTT assay and Annexin V/PI flow cytometry. Dose‑response curves demonstrated that the 20 mm nozzle induced significantly higher cytotoxicity: after 120 seconds, U87 viability dropped by 78 % and MDA‑MB‑231 by 71 %, whereas the 60 mm nozzle achieved only 55 % and 48 % reductions, respectively. The addition of ROS scavenger N‑acetylcysteine or NO scavenger L‑arginine rescued a large fraction of cells, confirming that reactive oxygen and nitrogen species (RONS) are the primary mediators of cell death. Importantly, both cell lines responded similarly despite differing p53 status, indicating that µCAP’s anti‑cancer effect is not tumor‑type specific.
The authors conclude that nozzle length is a critical design parameter that modulates electric field strength, electron density, and consequently the yield of biologically active RONS. The 20 mm µCAP delivers a concentrated burst of short‑lived radicals and long‑lived oxidants, producing rapid, dose‑dependent tumor cell killing without reliance on specific genetic vulnerabilities. This work establishes µCAP as a versatile, portable plasma platform with potential for targeted cancer therapy. Future directions include in‑vivo safety profiling, tissue penetration studies, and integration of real‑time RONS monitoring to refine treatment dosimetry and expand clinical applicability.
Comments & Academic Discussion
Loading comments...
Leave a Comment