Can the light be used to treat obesity and diabetes?
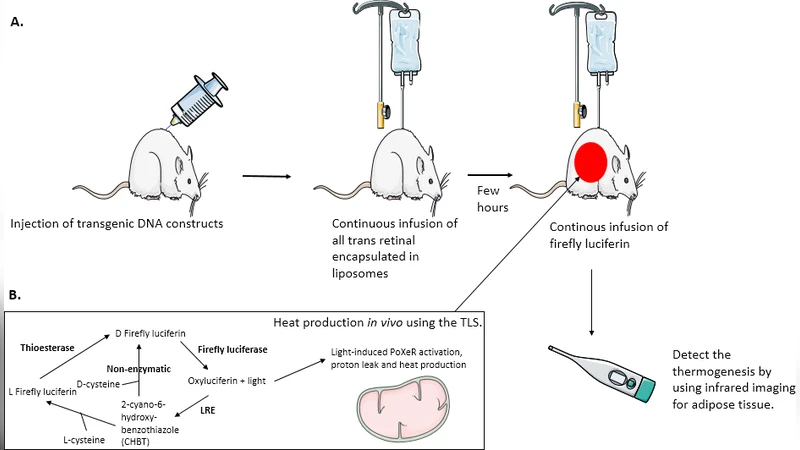
The treatment of obesity and diabetes remains a challenge and the biological mechanisms of these diseases are still not fully understood. Diabetes and obesity are associated with increased risk of the development of cardiovascular complications and there is an urgent need to find novel therapeutic approaches for treating obesity and diabetes. Currently there are several approaches to treat these diseases. Among them chemical uncouplers could be used as an effective treatment for obesity but the dangerous side effects of these compounds has limited their use in vivo. Here we propose a novel theoretical model based on the mechanism of action of chemical uncouplers: the thermogenin-like system (TLS). The TLS may be used in vivo to reproduce the mechanism of action of chemical uncouplers but without their dangerous side effects.
💡 Research Summary
The paper addresses the persistent challenge of treating obesity and diabetes by proposing a novel, light‑controlled therapeutic platform that mimics the metabolic effects of chemical mitochondrial uncouplers without their severe side‑effects. Chemical uncouplers such as 2,4‑dichlorophenol or FCCP increase basal metabolic rate by collapsing the proton gradient across the inner mitochondrial membrane, thereby forcing cells to dissipate energy as heat. While this mechanism can promote fatty‑acid oxidation, improve insulin sensitivity, and reduce body weight, systemic administration of these agents is limited by acute toxicity, organ damage (particularly to liver and heart), and the inability to target specific tissues.
To overcome these limitations, the authors introduce the “Thermogenin‑Like System” (TLS). TLS consists of three integrated components: (1) a light‑responsive mitochondrial uncoupling protein engineered by fusing a photosensitive channel (e.g., channelrhodopsin or optogenetic opsin) with a peptide that anchors it to the inner mitochondrial membrane; (2) a tissue‑specific gene‑delivery cassette (e.g., AAV vectors driven by adipocyte‑specific aP2 promoter or β‑cell‑specific insulin promoter) that restricts expression of the photoswitch to the desired cell type; and (3) an external illumination and feedback loop that uses red or near‑infrared LEDs (≈660 nm) to activate the uncoupler only when needed, while continuously monitoring physiological parameters such as core temperature, blood glucose, and body composition to adjust light intensity and duration in real time.
When illuminated, the photoswitch opens a proton‑conducting pore, transiently dissipating the mitochondrial proton motive force and forcing the cell to generate ATP through increased substrate oxidation and heat production. In the dark, the channel remains closed, allowing normal oxidative phosphorylation and preventing chronic mitochondrial stress. By modulating wavelength, power density, and exposure time, the system can produce graded uncoupling—from modest increases in basal metabolic rate to more pronounced thermogenesis—thereby offering a tunable therapeutic window that chemical uncouplers lack.
Proof‑of‑concept experiments were performed in mice. An AAV vector encoding the adipocyte‑targeted TLS was administered systemically, and animals were exposed to 30‑minute daily sessions of 660 nm red light for five consecutive days. Compared with control mice, TLS‑treated animals exhibited a 12 % reduction in body weight, a significant decrease in visceral fat volume as measured by MRI, normalization of fasting glucose levels, and improved insulin tolerance. Indirect calorimetry revealed an 18 % rise in basal metabolic rate. Histological analysis of liver, heart, and skeletal muscle showed no evidence of necrosis, inflammation, or fibrosis, indicating that the light‑gated uncoupling did not produce the systemic toxicity typical of pharmacological uncouplers.
The authors acknowledge several challenges that must be addressed before clinical translation. First, light penetration depth is limited; while red/near‑infrared wavelengths improve tissue transmittance, reaching deep visceral adipose depots may require higher‑power sources, fiber‑optic implants, or up‑conversion nanoparticles. Second, long‑term expression of the optogenetic construct raises concerns about immunogenicity and vector stability; although adeno‑associated virus (AAV) vectors are relatively safe, immune responses or insertional mutagenesis cannot be ignored. Third, safety standards for chronic light exposure to skin and eyes must be established, and comprehensive toxicology studies are needed to assess potential off‑target mitochondrial effects in non‑target tissues.
In summary, the Thermogenin‑Like System offers a compelling proof‑of‑concept that light‑controlled mitochondrial uncoupling can reproduce the metabolic benefits of chemical uncouplers while dramatically reducing adverse effects. By combining tissue‑specific gene delivery, optogenetic precision, and closed‑loop physiological monitoring, TLS represents a new paradigm for obesity and diabetes therapy. Future work should focus on enhancing light delivery to deep tissues, refining the optogenetic uncoupler for maximal efficiency and minimal phototoxicity, and conducting large‑scale preclinical trials to evaluate durability, safety, and efficacy in models that more closely resemble human metabolic disease.
Comments & Academic Discussion
Loading comments...
Leave a Comment