Drying-Induced Atomic Structural Rearrangements in Sodium-Based Calcium-Alumino-Silicate-Hydrate Gel and the Mitigating Effects of ZrO$_2$ Nanoparticles
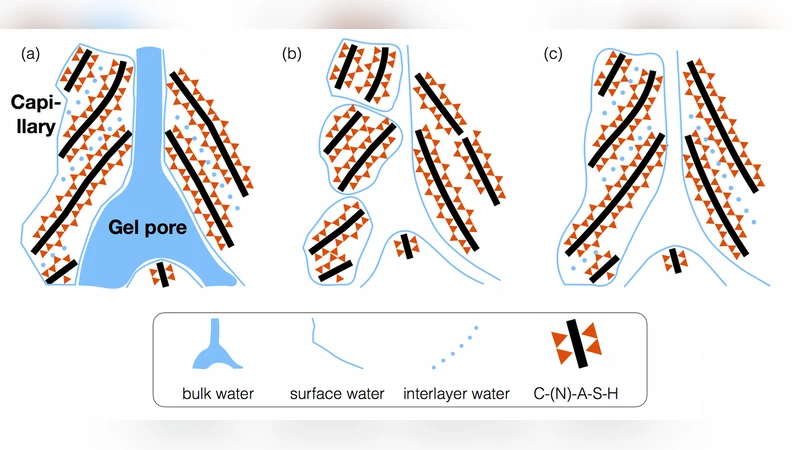
Conventional drying of colloidal materials and gels (including cement) can lead to detrimental effects due to the buildup of internal stresses as water evaporates from the nano/microscopic pores. However, the underlying nanoscopic alterations in these gel materials that are, in part, responsible for macroscopically-measured strain values, especially at low relative humidity, remain a topic of open debate in the literature. In this study, sodium-based calcium-alumino-silicate-hydrate (C-(N)-A-S-H) gel, the major binding phase of silicate-activated blast furnace slag (one type of low-CO$_2$ cement), is investigated from a drying perspective, since it is known to suffer extensively from drying-induced microcracking. By employing in situ synchrotron X-ray total scattering measurements and pair distribution function (PDF) analysis we show that the significant contributing factor to the strain development in this material at extremely low relative humidity (0%) is the local atomic structural rearrangement of the C-(N)-A-S-H gel, including collapse of interlayer spacing and slight disintegration of the gel. Moreover, analysis of the medium range (1.0 - 2.2 nm) ordering in the PDF data reveals that the PDF-derived strain values are in much closer agreement (same order of magnitude) with the macroscopically measured strain data, compared to previous results based on reciprocal space X-ray diffraction data. From a mitigation standpoint, we show that small amounts of ZrO$_2$ nanoparticles are able to actively reinforce the structure of silicate-activated slag during drying, preventing atomic level strains from developing. Mechanistically, these nanoparticles induce growth of a silica-rich gel during drying, which, via density functional theory calculations, we show is attributed to the high surface reactivity of tetragonal ZrO$_2$.
💡 Research Summary
This paper investigates the drying‑induced structural changes of sodium‑based calcium‑alumino‑silicate‑hydrate (C‑(N)‑A‑S‑H) gel, the principal binding phase in silicate‑activated blast‑furnace slag, and evaluates the mitigating effect of zirconia (ZrO₂) nanoparticles. Conventional drying of cementitious gels leads to internal stresses and microcracking, especially at low relative humidity (RH), yet the nanoscopic mechanisms that translate into macroscopic strain have remained ambiguous.
Using in‑situ synchrotron X‑ray total‑scattering, the authors collected pair‑distribution‑function (PDF) data while progressively lowering RH from 100 % to 0 %. At 0 % RH the PDF shows a pronounced reduction in the peak associated with the interlayer spacing (~0.9 nm) and a general loss of intensity at low‑Q, indicating collapse of the interlayer distance and partial disintegration of the gel network. Medium‑range analysis (1.0–2.2 nm) reveals that the PDF‑derived strain closely matches the strain measured macroscopically (e.g., by digital image correlation), confirming that atomic‑scale rearrangements are the dominant source of macroscopic deformation. This contrasts with earlier studies that relied on conventional X‑ray diffraction, which underestimated strain because diffraction peaks are broadened or absent in the highly disordered C‑(N)‑A‑S‑H.
To explore mitigation, the authors introduced 0.5 wt % tetragonal ZrO₂ nanoparticles into the gel. During the same drying protocol, the ZrO₂‑modified system retained its interlayer peak intensity and displayed markedly lower medium‑range strain. PDF data suggest that the nanoparticles promote the formation of a silica‑rich gel phase during drying, effectively “healing” the collapsing layers. Density‑functional‑theory (DFT) calculations support this mechanism: the high surface reactivity of tetragonal ZrO₂ (oxygen vacancies and strong Lewis acidity) adsorbs water and silicate species, facilitating Si–O–Si bond regeneration and the nucleation of a Si‑rich gel that bonds to the existing C‑(N)‑A‑S‑H matrix. This additional phase increases local density and prevents the collapse of interlayer spacing, thereby suppressing atomic‑level strain.
Key contributions of the work are: (1) direct experimental evidence that drying‑induced interlayer collapse and modest gel disintegration drive macroscopic strain in low‑CO₂ cement systems; (2) demonstration that PDF analysis provides a more accurate quantification of strain in amorphous or nanocrystalline gels than traditional diffraction; (3) identification of ZrO₂ nanoparticles as an active nanoreinforcement that triggers in‑situ silica‑rich gel growth, with a mechanistic explanation grounded in DFT surface chemistry.
The authors suggest that future research should compare other high‑surface‑energy oxides (e.g., TiO₂, Al₂O₃), examine the influence of nanoparticle size and morphology, and validate the approach in real concrete specimens under long‑term drying and service conditions. By bridging atomic‑scale observations with macroscopic performance, this study offers a promising pathway to enhance the durability of low‑carbon cementitious materials.
Comments & Academic Discussion
Loading comments...
Leave a Comment