Quantitative analysis of the potential role of basal cell hyperplasia in the relationship between clonal expansion and radon concentration
Applying the two-stage clonal expansion model to epidemiology of lung cancer among uranium miners, it has been revealed that radon acts as a promoting agent facilitating the clonal expansion of already mutated cells. Clonal expansion rate increases non-linearly by radon concentration showing a plateau above a given exposure rate. The underlying mechanisms remain unclear. Earlier we proposed that progenitor cell hyperplasia may be induced upon chronic radon exposure. The objective of the present study is to test whether the induction of hyperplasia may provide a quantitative explanation for the plateau in clonal expansion rate. For this purpose, numerical epithelium models were prepared with different number of basal cells. Cell nucleus hits were computed by an own-developed Monte-Carlo code. Surviving fractions were estimated based on the number of cell nucleus hits. Cell division rate was computed supposing equilibrium between cell death and cell division. It was also supposed that clonal expansion rate is proportional to cell division rate, and therefore the relative increase in cell division rate and clonal expansion rate are the same functions of exposure rate. While the simulation results highly depend on model parameters with high uncertainty, a parameter set has been found resulting in a cell division rate exposure rate relationship corresponding to the plateau in clonal expansion rate. Due to the high uncertainty of the applied parameters, however, further studies are required to decide whether the induction of hyperplasia is responsible for the non-linear increase in clonal expansion rate or not. Nevertheless the present study exemplifies how computational modelling can contribute to the integration of observational and experimental radiation protection research.
💡 Research Summary
The authors address a puzzling feature of radon‑induced lung carcinogenesis: epidemiological analyses using a two‑stage clonal expansion (TSCE) model show that radon acts as a promoter, increasing the clonal expansion rate of already mutated cells in a non‑linear fashion that plateaus at exposure rates above roughly 150 WLM yr⁻¹. The biological basis of this plateau has not been identified. Building on earlier suggestions that chronic radon exposure may induce hyperplasia of bronchial basal (progenitor) cells, the present study asks whether such hyperplasia can quantitatively reproduce the observed exposure‑response curve of clonal expansion.
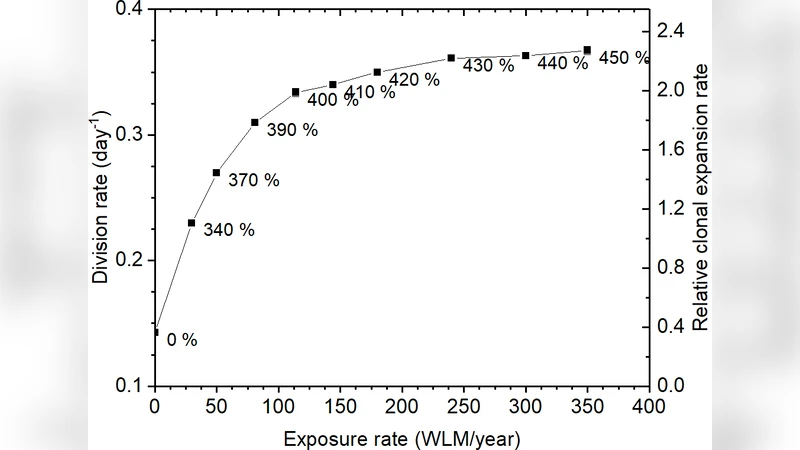
A key assumption is that the clonal expansion rate (γ) is proportional to the basal cell division rate (α). Consequently, the functional form of γ(D) derived from epidemiology (Equation 1) must be identical to that of α(D) (Equation 2). The authors therefore construct a series of computational tissue models in which the number of basal cells is increased by 0 % to 450 % (Table 1), thereby thickening the epithelium while keeping the overall surface area constant. For each hyperplasia level they simulate alpha‑particle tracks from radon progeny using a custom Monte‑Carlo code. The code incorporates the experimentally measured proportion of 218 Po (10.4 %) and 214 Po (89.6 %) decays, isotropic particle directions, SRIM‑derived ranges, and a mucus layer (baseline 11 µm, also examined at 22 µm) in which decay density decays exponentially with a half‑value thickness of 6 µm.
Cellular survival is modeled as an exponential function of the number of nuclear hits, p_sv = exp(−β n) with β = 0.285, applied equally to basal and non‑basal cells. The spontaneous death rate of non‑basal cells is fixed (17 100 mm⁻²) and the basal cell division rate in unexposed tissue is allowed to vary between 1/7 day⁻¹ and 1/100 day⁻¹, reflecting the wide range reported in the literature. Assuming steady‑state equilibrium between cell loss (from radiation‑induced death) and cell replacement (by division of basal cells), the model yields an exposure‑dependent division rate α(D).
Simulation results are presented separately for high‑dose “hot spots” (the small bronchial regions where radon progeny deposition is maximal) and for the more typical low‑dose airway surface. In hot spots, achieving the α(D) curve prescribed by Equation 2 would require an implausibly large increase in basal cell number—often more than three‑ to four‑fold—even when the mucus layer is doubled. Such a degree of hyperplasia is biologically unlikely, suggesting that hyperplasia alone cannot explain the plateau in these highly irradiated zones.
Conversely, in the low‑dose regions, with a normal mucus thickness (11 µm) and a low baseline basal cell division rate (1/100 day⁻¹), the model finds reasonable agreement: a modest 150 %–250 % increase in basal cells suffices to reproduce the epidemiologically derived α(D) up to about 200 WLM yr⁻¹. At higher exposures (≈350 WLM yr⁻¹) only a 2.2‑fold increase is needed, and the relationship between hyperplasia and exposure remains sub‑linear, mirroring the observed flattening of the clonal expansion curve.
These findings demonstrate that, under certain plausible parameter sets, radon‑induced basal cell hyperplasia can generate a dose‑response for cell division that matches the TSCE‑derived clonal expansion curve, thereby offering a mechanistic explanation for the plateau. However, the outcome is highly sensitive to three poorly constrained variables: (i) the normal basal cell division rate, (ii) the thickness of the mucus layer (which can be altered by inflammation), and (iii) the exact spatial distribution of radon progeny deposition. Because these parameters span wide ranges in experimental reports, the authors caution that their conclusions remain tentative.
The paper concludes that computational tissue‑level modeling is a valuable bridge between epidemiological observations and radiation biology, but that definitive proof will require histological studies of former uranium miners’ airways to quantify basal cell numbers and mucus thickness across different airway regions. Such data would allow validation (or refutation) of the hyperplasia hypothesis and clarify whether it truly underlies the non‑linear, plateauing behavior of radon‑induced clonal expansion.
Comments & Academic Discussion
Loading comments...
Leave a Comment