Microfluidics for Chemical Synthesis: Flow Chemistry
Klavs F. Jensen is Warren K. Lewis Professor in Chemical Engineering and Materials Science and Engineering at the Massachusetts Institute of Technology. Here he describes the use of microfluidics for chemical synthesis, from the early demonstration examples to the current efforts with automated droplet microfluidic screening and optimization techniques.
💡 Research Summary
Klavs F. Jensen’s paper provides a comprehensive overview of how microfluidic technology has reshaped chemical synthesis through flow chemistry. The author begins by outlining the inherent limitations of traditional batch processes—poor heat and mass transfer, safety hazards associated with exothermic reactions, and difficulties in scaling up. He then argues that microfluidic reactors, with channel dimensions on the order of tens to hundreds of micrometers, exploit laminar flow conditions to achieve precise control over residence time, mixing, and temperature, thereby overcoming many of these constraints.
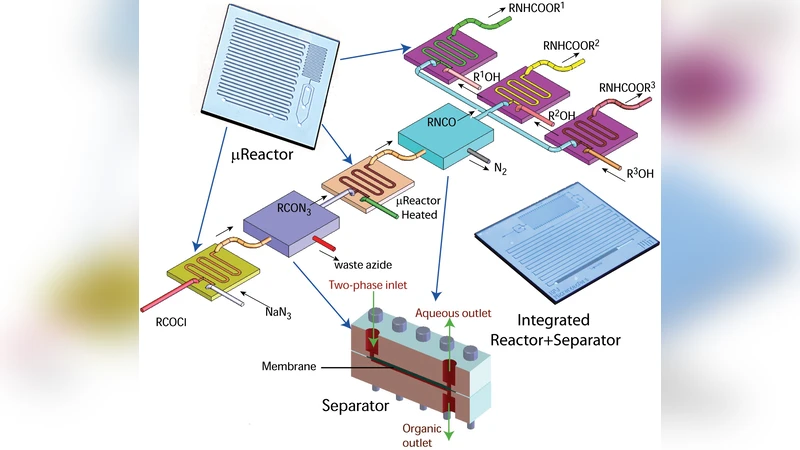
Early demonstrations, dating from the late 1990s to the early 2000s, are presented as proof‑of‑concept studies. In these experiments, simple oxidation, catalytic hydrogenation, and organic coupling reactions were performed in continuous flow with residence times reduced from hours to seconds. The high surface‑to‑volume ratio of the channels enabled rapid heat dissipation, allowing highly exothermic reactions to be conducted safely and with markedly improved selectivity and yield (often exceeding 90 %).
The paper then delves into the engineering fundamentals of microfluidic reactors. It discusses the dominance of diffusion‑driven mixing in laminar flow, the design of various micromixers (split‑and‑recombine, serpentine, staggered herringbone), and the integration of on‑chip temperature sensors and external heaters/coolers to create closed‑loop feedback control. These features permit the construction of multi‑step flow sequences where intermediates are generated, transformed, and purified in a single uninterrupted stream, effectively collapsing what would be a multi‑batch process into a continuous operation.
A major focus is placed on droplet‑based microfluidics, where discrete aqueous or organic droplets are generated within an immiscible carrier fluid. Droplet reactors act as isolated micro‑reactors, each capable of housing a unique set of reaction conditions. By leveraging high‑throughput droplet generation (up to tens of thousands per hour) and automated liquid handling robotics, the author demonstrates rapid combinatorial screening of catalyst compositions, solvent systems, and temperature profiles. Machine‑learning algorithms are employed to analyze the resulting data streams, iteratively suggesting new experimental points and converging on optimal conditions with far fewer experiments than traditional design‑of‑experiments approaches.
Integration with real‑time analytical tools is another cornerstone of the presented workflow. Inline Fourier‑transform infrared (FTIR), mass spectrometry (MS), nuclear magnetic resonance (NMR), and Raman spectroscopy provide instantaneous feedback on conversion, selectivity, and by‑product formation. This enables dynamic adjustment of flow rates, temperatures, and reagent concentrations during a run, effectively turning the microfluidic platform into a self‑optimizing reactor. The author cites case studies where such closed‑loop control led to a 3‑fold increase in product yield and a dramatic reduction in impurity formation compared with open‑loop batch processes.
Finally, Jensen addresses the remaining challenges that must be tackled for widespread adoption. Channel fouling, limited chemical compatibility of common microfabrication materials (e.g., PDMS, glass), and the translation of microscale performance to industrial‑scale production are highlighted. Proposed solutions include the development of modular, parallelized channel arrays, the use of chemically resistant substrates (e.g., fluorinated polymers, metal‑lined silicon), and the establishment of standardized interfacing protocols for seamless integration with downstream purification units. The paper also envisions a future where cloud‑based data repositories and AI‑driven reactor design tools democratize access to microfluidic synthesis, paving the way for a “digital chemistry factory” that can rapidly prototype, optimize, and scale new chemical entities.
In summary, Jensen’s work demonstrates that microfluidic flow chemistry not only offers superior reaction control, safety, and efficiency but also serves as a platform for automation, high‑throughput experimentation, and real‑time optimization. By coupling these capabilities with advanced analytics and machine learning, the field is poised to transform the way chemists develop and manufacture chemicals, moving from empirical batch experimentation toward a predictive, data‑driven paradigm.
Comments & Academic Discussion
Loading comments...
Leave a Comment