Tensile rupture of medial arterial tissue studied by X-ray micro-tomography on stained samples
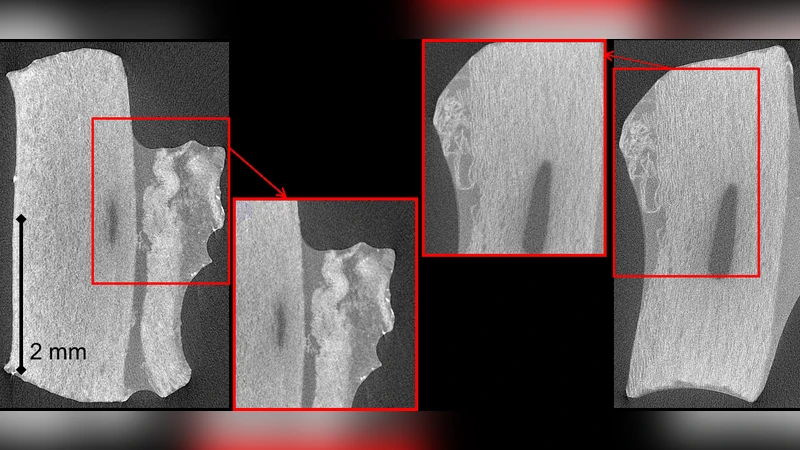
Detailed characterization of damage and rupture mechanics of arteries is one the current challenges in vascular biomechanics, which requires developing suitable experimental approaches. This paper introduces an approach using in situ tensile tests in an X-ray micro-tomography setup to observe mechanisms of damage initiation and progression in medial layers of porcine aortic samples. The technique requires the use of sodium polytungstate as a contrast agent, of which the conditions for use are detailed in this paper. Immersion of the samples during 24 hours in a 15 g.L-1 concentrated solution provided the best compromise for viewing musculo-elastic units in this tissue. The process of damage initiation, delamination and rupture of medial tissue under tensile loading was observed and can be described as an elementary process repeating several times until complete failure. This elementary process initiates with a sudden mode I fracture of a group of musculo-elastic units, followed by an elastic recoil of these units, causing mode II separation of these, hence a delamination plane. The presented experimental approach constitutes a basis for observation of other constituents, or for investigations on other tissues and damage mechanisms.
💡 Research Summary
This paper presents a novel experimental platform that combines in‑situ tensile testing with X‑ray micro‑computed tomography (micro‑CT) to directly visualize the damage initiation and rupture processes of the medial layer of porcine aorta. Because soft vascular tissue provides little X‑ray contrast, the authors systematically investigated the use of sodium polytungstate (Na₂WO₄·2H₂O) as a contrast agent. Immersing the arterial strips in a 15 g·L⁻¹ solution for 24 hours yielded optimal differentiation of the musculo‑elastic units (MUs) – the functional bundles of smooth‑muscle cells and elastic fibers – without excessively altering the mechanical properties of the tissue.
The experimental setup integrates a miniature tensile rig onto a micro‑CT scanner. Samples are gripped at both ends and stretched at a quasi‑static rate (<0.1 mm·s⁻¹) while the scanner rotates, acquiring a full 360° projection series every few seconds. Scanning parameters (80 kV, 100 µA, 10 µm voxel size) were chosen to balance spatial resolution with radiation dose, allowing continuous 3‑D imaging throughout the loading history. The resulting data set consists of synchronized load‑displacement curves and time‑resolved volumetric images.
Analysis of the reconstructed volumes revealed a repeatable “elementary process” that governs medial rupture. The first event is a sudden Mode I (opening) fracture that simultaneously breaks a cluster of MUs. This fracture creates a high‑energy release that causes the broken units to recoil elastically. The recoil generates shear stresses at the interface between the fractured and adjacent intact MUs, leading to a Mode II (shear) separation that forms a delamination plane. This sequence – Mode I fracture → elastic recoil → Mode II delamination – repeats several times, each iteration expanding the damaged zone until the specimen finally fails. Quantitative metrics such as the increase in fracture surface area, the drop in load, and the local strain fields were extracted for each cycle, providing insight into the energy dissipation and progression rate of the damage.
The authors also applied a Markov random‑field segmentation algorithm to automatically delineate individual MUs and to track their volumetric changes throughout loading. Correlating these morphological changes with the mechanical data demonstrated that regions of high curvature and reduced MU volume are precursors to fracture, suggesting a possible predictive marker for rupture in more complex, diseased vessels.
Key contributions of the study include: (1) a validated contrast‑enhancement protocol for soft vascular tissue in micro‑CT; (2) a fully synchronized mechanical‑imaging system that captures 3‑D structural evolution in real time; (3) the identification of a distinct fracture‑delamination mechanism that differs from traditional fiber‑breakage models; and (4) a quantitative dataset that can be directly incorporated into finite‑element models of arterial failure.
Limitations are acknowledged. Radiation exposure may subtly modify tissue stiffness, the sample size is restricted to ≤5 mm specimens, and the experiments are performed ex‑vivo, lacking physiological pressure and flow conditions. Future work is proposed to reduce dose (e.g., phase‑contrast imaging), to test diseased or pharmacologically treated arteries, and to integrate the methodology with in‑vivo imaging modalities.
In conclusion, this work establishes a powerful, non‑destructive approach for observing vascular damage at the microscale. By revealing the stepwise Mode I‑to‑Mode II transition that underlies medial rupture, the study provides a mechanistic foundation for improved computational models, risk assessment of aneurysm or plaque rupture, and the development of targeted therapeutic strategies.
Comments & Academic Discussion
Loading comments...
Leave a Comment