We report a gas eruption phenomenon caused by electrolysis of liquid Ga-In alloy in an electrolyte, especially NaOH solution. A volcanic eruption-like blowout of gas occurred from the orifice on the alloy surface. In addition to gas plume, large gas bubbles were also generated and the total gas yield increased as In ratio was increased. It is found that destructiveness of the passivation layer on the Ga-In alloy is critical to gas generation. The mechanism of gas eruption can be ascribed to a galvanic interaction happens owing to passivation film and alloy with different activity connected as electrode in electrolyte. Further investigation demonstrated that the lattice of the film expands because of the incorporation of indium, which brings about the decrease in band gap and finally enhances more gas generation. These findings regain the basic understanding of room temperature liquid metal inside electrolyte.
Deep Dive into Gas Eruption Phenomenon Happening from Ga-In Alloy in Electrolyte.
We report a gas eruption phenomenon caused by electrolysis of liquid Ga-In alloy in an electrolyte, especially NaOH solution. A volcanic eruption-like blowout of gas occurred from the orifice on the alloy surface. In addition to gas plume, large gas bubbles were also generated and the total gas yield increased as In ratio was increased. It is found that destructiveness of the passivation layer on the Ga-In alloy is critical to gas generation. The mechanism of gas eruption can be ascribed to a galvanic interaction happens owing to passivation film and alloy with different activity connected as electrode in electrolyte. Further investigation demonstrated that the lattice of the film expands because of the incorporation of indium, which brings about the decrease in band gap and finally enhances more gas generation. These findings regain the basic understanding of room temperature liquid metal inside electrolyte.
1
Gas Eruption Phenomenon Happening from Ga-In Alloy in Electrolyte
Ruiqi Zhao,1,2 Hongzhang Wang,1,3 Jianbo Tang,1,3 Wei Rao,1,4,a) and Jing Liu,1,3,4,a)
1 Beijing Key Lab of CryoBiomedical Engineering and Key Lab of Cryogenics, Technical
Institute of Physics and Chemistry, Chinese Academy of Sciences, Beijing 100190, China
2Department of Chemistry, University of Chinese Academy of Sciences, Beijing 100049, China
3Department of Biomedical Engineering, School of Medicine, Tsinghua University, Beijing
100084, China
4College of Future Technology, University of Chinese Academy of Sciences, Beijing 100049,
China
a) Authors to whom correspondence should be addressed. E-mail: weirao@mail.ipc.ac.cn;
jliu@mail.ipc.ac.cn.
Abstract: We report a gas eruption phenomenon caused by electrolysis of liquid Ga-In alloy
in an electrolyte, especially NaOH solution. A volcanic eruption-like blowout of gas occurred
from the orifice on the alloy surface. In addition to gas plume, large gas bubbles were also
generated and the total gas yield increased as In ratio was increased. It is found that
destructiveness of the passivation layer on the Ga-In alloy is critical to gas generation. The
mechanism of gas eruption can be ascribed to a galvanic interaction happens owing to
passivation film and alloy with different activity connected as electrode in electrolyte. Further
investigation demonstrated that the lattice of the film expands because of the incorporation
of indium, which brings about the decrease in band gap and finally enhances more gas
generation. These findings regain the basic understanding of room temperature liquid metal
inside electrolyte.
Recently, studies on gallium-based liquid metals have drawn considerable attention,
particularly for their potential applications in hydrogen generation. Such alloys have in fact
been the focus of many studies. The addition of aluminum to Ga alloy contributes to
self-driven motions.1-7 The majority of studies have proven the addition of aluminum or other
metal particles as the main regulator of hydrogen generation.3, 8-14 The generation of gas is
considered to be a propelling force of a bunch of phenomena, including the motion of
Ga-In-Al in aqueous NaOH solutions,15 and the oscillation phenomenon of a copper wire
embedded inside Ga-In-Al self-powering system.16 Alternatively, Ga–In liquid metal can be
applied versatile to self-healing or contrast enhancement.17 Tang et al. showed that
hydrogen could be generated from a drop of Ga-In alloy when left in contact with solid metal
particles.18 However, phenomenon of gas evolution from bulk Ga-In alloy has always been
overlooked and no studies have yet been reported.
In this letter, we report an interesting gas eruption phenomenon occurring in Ga-In alloy
when it contacts with an electrolyte. Over the experiment, 10 cm petri dish was pre-filled with
50 ml Ga-In alloy in the open air [Fig.1(a)], then 30 ml NaOH electrolyte was transferred onto
the surface of alloy. When immersed in a 1 M alkaline electrolyte, a number of gas plumes
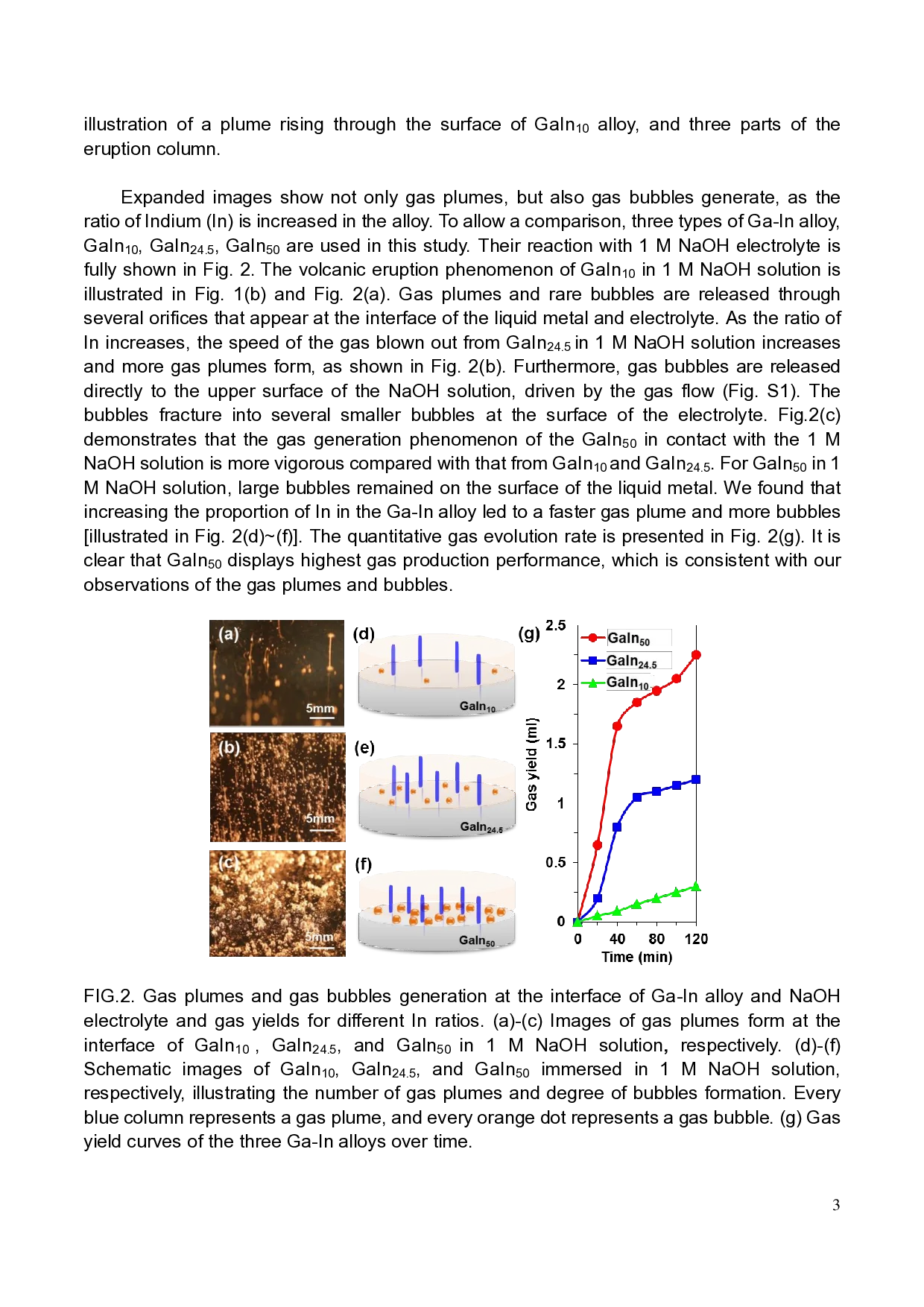
appeared at the surface of GaIn10 (Ga : In = 9:1,w/w, see Fig.1(b) & Multimedia view),
GaIn24.5 (Ga : In = 75.5:24.5, w/w, see Fig.S1& supplemental material S1) and GaIn50 alloy
(Ga : In =1:1, w/w, see Fig.1(c) & Multimedia view). The eruption phenomena have been
2
recorded with a Canon EOS 70D camera with macro lenses. The experiments were
performed at a temperature of 22°C.
The electrolyte activates the surface layer of Ga-In alloy and creates many orifices
[Fig.1(d), side view in supplemental material S2], from which gas column pours out. In
addition, the hydrogen gas generation is rapid and constant, and the continuous gas flow
forms a gas plume in the electrolyte (see Multimedia view). This is a similar phenomenon to
that of volatile gas blended with magma erupting from the seabed, which then rises to the
top of sea water. Considering the similarities with submarine volcanic eruptions, we divide
our eruption column into three zones19, namely regions of jet, ascent, and diffusion,
respectively [Fig.1(e)]. The jet region is located at the lowest part of the eruption column, and
the column width is approximately 50~200 μm. The gas flow from the orifice is sufficiently
powerful to overcome the frictional resistance, and the velocity at the alloy/electrolyte
interface is controlled mainly by the volatile gas release of the Ga-In alloy. In the ascent
region (upper zone), the speed of the gas column is slower than that in the jet phase. The
gas in the column scatters outward, and the force driving the upward motion is dominated by
buoyancy. The diffusion region is located at the top of the eruption column where the
eruption column rises and spreads in the horizontal direction. In diffusion region, the
pressure of the eruption column and the external atmosphere reaches equilibrium.
FIG.1. Images of gas plumes and bubbles appearing on the
…(Full text truncated)…
This content is AI-processed based on ArXiv data.