Multi-task Neural Networks for Personalized Pain Recognition from Physiological Signals
Pain is a complex and subjective experience that poses a number of measurement challenges. While self-report by the patient is viewed as the gold standard of pain assessment, this approach fails when patients cannot verbally communicate pain intensity or lack normal mental abilities. Here, we present a pain intensity measurement method based on physiological signals. Specifically, we implement a multi-task learning approach based on neural networks that accounts for individual differences in pain responses while still leveraging data from across the population. We test our method in a dataset containing multi-modal physiological responses to nociceptive pain.
💡 Research Summary
Pain assessment traditionally relies on self‑report scales such as the Numerical Rating Scale or Visual Analogue Scale, which become unusable when patients cannot communicate verbally or lack cognitive capacity. This paper tackles the problem by developing an objective, wearable‑friendly method that estimates pain intensity solely from physiological signals—specifically skin conductance (SC) and electrocardiogram (ECG) derived heart‑rate variability (HRV). The authors argue that these two modalities capture complementary aspects of the autonomic nervous system (sympathetic and parasympathetic activity) and can be measured with wrist‑mounted sensors, making the approach suitable for continuous, real‑time monitoring in clinical settings.
The study uses the publicly available BioVid Heat Pain dataset, which contains recordings from 87 healthy participants subjected to calibrated thermal stimuli at five levels (baseline T0 and four pain levels T1‑T4). Each stimulus lasts four seconds, followed by a randomized recovery period, and the authors extract 5.5‑second windows starting one second after the target temperature is reached. This yields 8,700 samples (20 per class per subject). From each window, a set of handcrafted features is computed. For SC, twelve statistical and temporal descriptors are extracted, including maximum, range, standard deviation, inter‑quartile range, root‑mean‑square, mean, mean absolute first‑ and second‑order differences (both raw and standardized), skewness, and kurtosis. For ECG, five HRV‑related features are derived: mean inter‑beat interval (IBI), root‑mean‑square of successive differences (RMSSD), standard deviation of NN intervals (SDNN), slope of a linear regression fitted to the IBI series, and the ratio SDNN/RMSSD. All features are standardized before modeling.
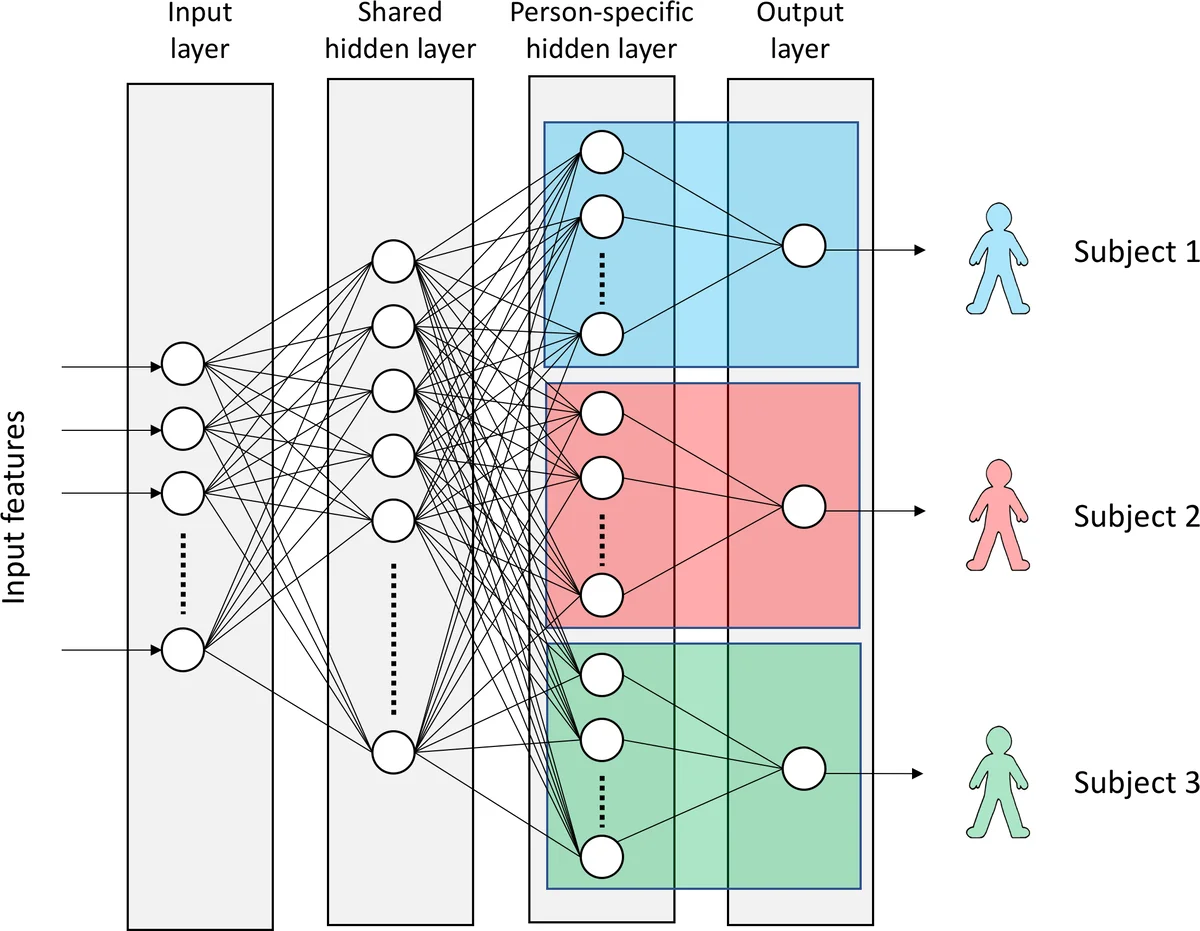
The core methodological contribution is a multi‑task learning (MTL) neural network with hard parameter sharing. The network consists of an input layer, a shared hidden layer common to all subjects, and a subject‑specific hidden layer that feeds into a sigmoid output unit for binary classification (pain vs. no‑pain or pain level vs. baseline). This architecture allows the model to learn population‑wide representations while simultaneously adapting to individual physiological response patterns. The authors employ ReLU activations, dropout, weight‑norm constraints, and early stopping to regularize training. Implementation is done in TensorFlow 1.2.1 and Keras 2.0.6.
For evaluation, the authors compare four baseline classifiers—logistic regression (LR), linear SVM, RBF‑kernel SVM, and a single‑task neural network (ST‑NN)—against the proposed MTL‑NN. Ten‑fold cross‑validation is used throughout. Results show that SC features alone outperform ECG features across all models, confirming the stronger pain‑related signal in skin conductance. When both modalities are combined, the MTL‑NN consistently achieves the highest accuracies. Notably, for the most intense pain level (P4) the MTL‑NN reaches 82.75 % accuracy, and for the next level (P3) 70.04 %, surpassing prior work that reported 74.1 % and 65.0 % respectively (the prior work also used EMG in addition to SC and ECG). For lower pain levels (P1, P2) the gains are modest, indicating that discriminating subtle pain intensities remains challenging with the current feature set.
The paper’s contributions can be summarized as follows: (1) Demonstration that a lightweight, wearable‑compatible sensor suite (SC + ECG) can provide sufficient information for reliable pain intensity classification; (2) Introduction of a hard‑shared MTL neural network that personalizes predictions without sacrificing the benefits of pooled data; (3) Empirical evidence that this approach outperforms traditional linear models and single‑task deep learning baselines on a standard benchmark.
Limitations are acknowledged. The dataset consists solely of healthy volunteers in a controlled laboratory environment, so external validity to postoperative patients, intensive‑care units, or chronic pain populations is not established. Moreover, the exclusion of electromyography (EMG) and other biosignals may limit the ceiling performance achievable.
Future work outlined by the authors includes: refining task definitions using meta‑information (e.g., gender, age) and similarity‑based clustering to improve parameter sharing; exploring more sophisticated time‑series deep learning architectures such as CNN‑LSTM or variational autoencoders for automated feature learning; and translating the algorithm to a real‑time wearable device to assess latency, power consumption, and robustness in clinical workflows. If these directions succeed, the proposed system could become a practical tool for continuous, objective pain monitoring, aiding clinicians in pain management decisions where self‑report is unavailable.
Comments & Academic Discussion
Loading comments...
Leave a Comment