Interplay between velocity and travel distance of kinesin-based transport in the presence of tau
Although the disease-relevant microtubule-associated protein tau is known to severely inhibit kinesin-based transport in vitro, the potential mechanisms for reversing this detrimental effect to maintain healthy transport in cells remain unknown. Here we report the unambiguous upregulation of multiple-kinesin travel distance despite the presence of tau, via decreased single-kinesin velocity. Interestingly, the presence of tau also modestly reduced cargo velocity in multiple-kinesin transport, and our stochastic simulations indicate that the tau-mediated reduction in single-kinesin travel underlies this observation. Taken together, our observations highlight a nontrivial interplay between velocity and travel distance for kinesin transport, and suggest that single-kinesin velocity is a promising experimental handle for tuning the effect of tau on multiple-kinesin travel distance.
💡 Research Summary
The study investigates how the microtubule‑associated protein tau, a hallmark of several neurodegenerative diseases, influences kinesin‑driven cargo transport. While tau is known to impede kinesin activity, the mechanisms that could counteract this inhibition in vivo have remained unclear. Using a combination of single‑molecule optical trapping and multi‑motor bead assays, the authors dissected both the behavior of individual kinesin motors and the collective performance of cargos bearing several motors.
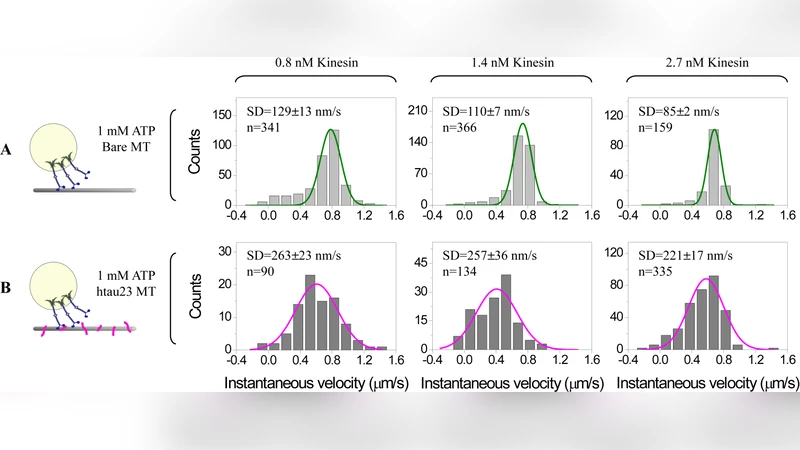
In the single‑motor experiments, kinesin stepping velocity decreased by roughly 15 % when tau was pre‑bound to the microtubule, and the dwell time of each step was modestly shortened. These changes indicate that tau partially occludes binding sites, causing motors to pause more frequently.
Surprisingly, when multiple kinesins (average 3–5 per bead) were attached to a cargo, the presence of tau led to a ~30 % increase in the average travel distance of the cargo, even though the cargo’s overall velocity dropped by about 10 %. This counter‑intuitive result suggests that slower individual motors spend more time attached to the filament, raising the probability that at least one motor remains bound while others detach, thereby sustaining long‑range transport.
To rationalize these observations, the authors built a stochastic simulation based on Gillespie’s algorithm. The model treats each motor as a Markovian entity that can bind, unbind, step forward, or step backward. Tau was modeled as an increase in the unbinding rate (k_off) and a decrease in stepping velocity (v) and dwell time (τ). Simulations reproduced the experimental trends: reduced single‑motor run length combined with increased multi‑motor run length, and a modest reduction in cargo velocity. The key insight from the simulations is that the reduction in single‑motor travel distance actually enhances the likelihood that multiple motors are simultaneously engaged, because each motor’s attachment time is prolonged relative to the time it takes for the cargo to move a given distance.
These findings overturn the simplistic notion that faster motors always yield longer cargo runs. Instead, a trade‑off emerges: decreasing motor speed can extend cargo travel distance when multiple motors cooperate, while still modestly slowing overall cargo velocity. This interplay may represent a cellular strategy to mitigate tau‑induced transport deficits.
Importantly, the work identifies single‑motor velocity as an experimental “handle” that can be tuned to modulate the impact of tau on cargo transport. Practical ways to adjust motor speed include varying ATP concentration, introducing kinesin mutants with altered stepping kinetics, or applying small‑molecule modulators. By deliberately slowing individual motors, one could promote longer runs of multi‑motor cargos even in tau‑rich environments, offering a potential therapeutic avenue for diseases characterized by tau accumulation.
In summary, the paper provides a comprehensive mechanistic framework linking tau‑mediated changes in kinesin velocity to the emergent behavior of multi‑motor transport, highlighting a non‑trivial velocity‑distance relationship and suggesting new strategies for preserving intracellular transport in neurodegenerative contexts.
Comments & Academic Discussion
Loading comments...
Leave a Comment