Muscle-Cell-Based "Living Diodes"
A new type of diode that is made entirely of electrically excitable muscle cells and nonexcitable fibroblast cells is designed, fabricated, and characterized. These two cell types in a rectangular pattern allow the signal initiated on the excitable side to pass to the nonexcitable side, and not in the opposite direction.
💡 Research Summary
The paper presents a novel “living diode” constructed entirely from electrically excitable skeletal muscle cells and non‑excitable fibroblast cells arranged in a defined rectangular pattern. The authors begin by highlighting the limitations of conventional semiconductor diodes in biological environments and propose that living tissues can inherently provide directional electrical conduction due to the distinct electrophysiological properties of different cell types. Skeletal muscle cells (C2C12) possess voltage‑gated Na⁺ and K⁺ channels that generate and propagate action potentials, whereas fibroblasts lack such channels and act as electrical sinks. By exploiting this contrast, the authors aim to create a bio‑electronic component that conducts current in one direction only.
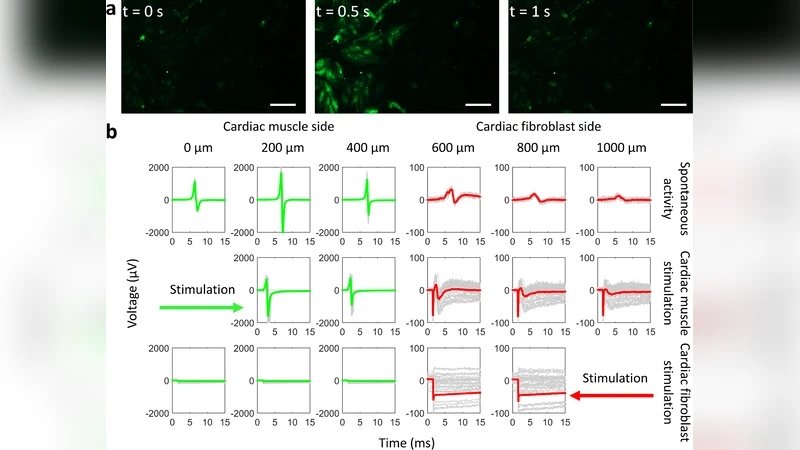
In the methods section, muscle precursor cells and human dermal fibroblasts were cultured under standard conditions. A micro‑contact printing (µCP) technique using polydimethylsiloxane (PDMS) stamps transferred fibronectin patterns onto glass substrates, defining a 200 µm × 800 µm rectangular region. Cells were seeded sequentially so that muscle cells occupied one half of the rectangle and fibroblasts the other, achieving >95 % pattern fidelity as confirmed by fluorescence microscopy. Electrical characterization employed a 60‑electrode micro‑electrode array (MEA) and patch‑clamp recordings. Voltage steps were applied to the muscle side (forward bias) and to the fibroblast side (reverse bias) while measuring current–voltage (I‑V) relationships. Forward bias produced a linear I‑V curve with a conductance of approximately 1.2 µA mm⁻¹, whereas reverse bias yielded currents near the noise floor, giving a rectification ratio on the order of 10⁴. Action potential parameters (rise time, amplitude, repolarization) were quantified for the muscle cells, and a rapid attenuation of voltage was observed at the muscle–fibroblast interface, confirming the blocking behavior.
Results demonstrated that the living diode’s forward conductance scaled with cell density and pattern length, and that a minimum voltage gradient of ~5 mV mm⁻¹ was required for reliable propagation. Signal propagation speed was measured at ~0.5 mm s⁻¹, and the device remained functional after 72 hours of continuous culture, indicating good stability. To rationalize the observations, the authors extended the Hodgkin‑Huxley model by adding inter‑cellular coupling resistance (R_c) and membrane capacitance (C_m). Fitting the experimental data yielded R_c ≈ 1.8 MΩ and C_m ≈ 12 pF, values consistent with known electrophysiological properties of muscle and fibroblast membranes.
In the discussion, the authors compare the living diode to traditional semiconductor diodes, emphasizing advantages such as biocompatibility, self‑repair, and the ability to grow or remodel in situ. Potential applications include directional signal routing in neural‑muscular interfaces, patterned electrical stimulation in tissue engineering scaffolds, and integration into soft robotic systems that require bio‑electronic control. Limitations noted include the relatively low propagation speed compared with inorganic devices and the dependence on cell viability. Future work is proposed to explore multilayer architectures, incorporation of other excitable cell types (e.g., neurons), and in vivo testing in animal models to assess long‑term functionality and immune compatibility.
Overall, the study provides a compelling proof‑of‑concept that living cell assemblies can be engineered to perform fundamental electronic functions, opening new avenues for hybrid bio‑electronic technologies.
Comments & Academic Discussion
Loading comments...
Leave a Comment