Antenna mechanism of length control of actin cables
Actin cables are linear cytoskeletal structures that serve as tracks for myosin-based intracellular transport of vesicles and organelles in both yeast and mammalian cells. In a yeast cell undergoing budding, cables are in constant dynamic turnover yet some cables grow from the bud neck toward the back of the mother cell until their length roughly equals the diameter of the mother cell. This raises the question: how is the length of these cables controlled? Here we describe a novel molecular mechanism for cable length control inspired by recent experimental observations in cells. This antenna mechanism involves three key proteins: formins, which polymerize actin, Smy1 proteins, which bind formins and inhibit actin polymerization, and myosin motors, which deliver Smy1 to formins, leading to a length-dependent actin polymerization rate. We compute the probability distribution of cable lengths as a function of several experimentally tuneable parameters such as the formin-binding affinity of Smy1 and the concentration of myosin motors delivering Smy1. These results provide testable predictions of the antenna mechanism of actin-cable length control.
💡 Research Summary
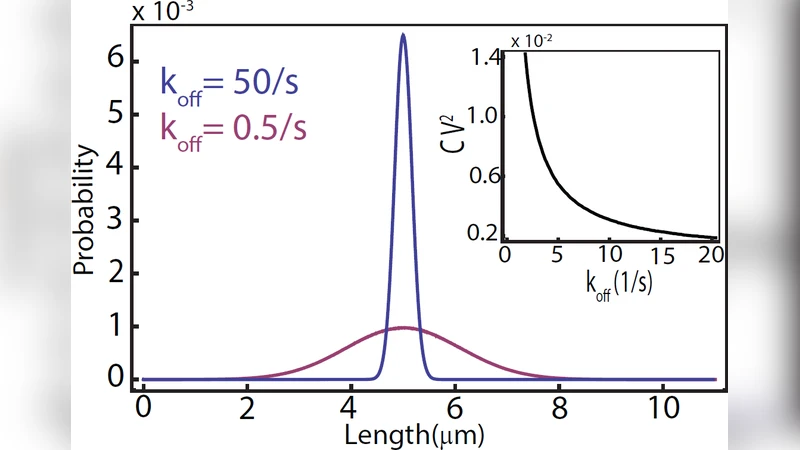
This paper addresses the long‑standing question of how actin cables in budding yeast attain a length that roughly matches the mother‑cell diameter despite continuous turnover. The authors propose an “antenna mechanism” in which three molecular players—formin, the formin‑binding inhibitor Smy1, and myosin motors that transport Smy1—interact to generate a length‑dependent polymerization rate. Formin catalyzes actin filament elongation, while Smy1 binds to formin and transiently blocks polymerization. Myosin motors walk along the cable, delivering Smy1 to the formin located at the cable tip; the longer the cable, the higher the probability that a myosin‑carried Smy1 reaches the tip within a given time interval. Consequently, the effective polymerization rate decreases with cable length, leading to a steady‑state length distribution.
The authors construct a stochastic master equation describing the binding and unbinding of Smy1 to formin (characterized by an affinity K_d and off‑rate k_off) and the delivery flux of Smy1, which scales with myosin concentration and motor velocity. By solving the equation analytically and confirming with Monte‑Carlo simulations, they obtain the probability distribution P(L) of cable lengths as a function of experimentally tunable parameters: Smy1‑formin affinity, Smy1 concentration, and myosin motor density. Parameter sweeps reveal that stronger Smy1‑formin binding or higher myosin delivery rates shift the distribution toward shorter mean lengths and reduce variance, whereas weaker binding or lower motor density permits longer, more variable cables. The model predicts a “length saturation” point where the inhibitory flux balances the polymerization flux, reproducing the observed plateau at the cell’s diameter.
Importantly, the paper outlines concrete experimental tests. Mutating the Smy1‑formin interaction site to alter K_d, overexpressing or depleting myosin V, and fluorescently tracking Smy1 delivery events would directly probe the predicted dependencies. The authors also discuss how the antenna concept could be generalized to other cytoskeletal structures that require size control, suggesting a broader principle where a filament itself acts as an antenna that senses its own length through motor‑mediated delivery of regulatory factors. In sum, the study provides a quantitative, testable framework for actin‑cable length regulation, bridging molecular biochemistry with cellular geometry.
Comments & Academic Discussion
Loading comments...
Leave a Comment