Chiral Interactions of Histidine in a Hydrated Vermiculite
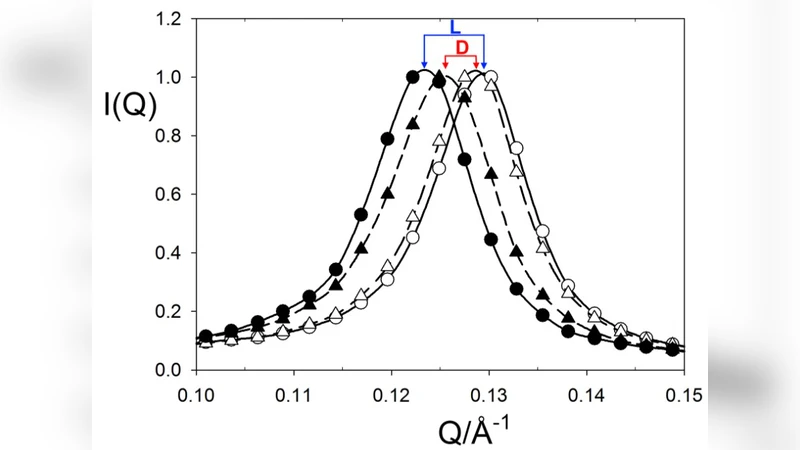
Recent work suggests a link between chiral asymmetry in the amino acid iso-valine extracted from the Murchison meteorite and the extent of hydrous alteration. We present the results of neutron scattering experiments on an exchanged, 1-dimensionally ordered n-propyl ammonium vermiculite clay. The vermiculite gel has a (001) d-spacing of order 5nm at the temperature and concentration of the experiments and the d-spacing responds sensitively to changes in concentration, temperature and electronic environment. The data show that isothermal addition of D-histidine or L-histidine solutions produces shifts in the d-spacing that are different for each enantiomer. This chiral specificity is of interest for the question of whether clays could have played an important role in the origin of biohomochirality.
💡 Research Summary
The paper investigates whether a layered clay mineral, vermiculite, can discriminate between the two enantiomers of the amino acid histidine, thereby offering a plausible inorganic mechanism for the emergence of biological homochirality. The authors prepared a one‑dimensional, ordered vermiculite gel by exchanging the interlayer cations with n‑propyl ammonium. In the hydrated state the (001) basal spacing (d‑spacing) is on the order of 5 nm, and this spacing is known to be highly sensitive to temperature, solute concentration, and the electronic environment of the interlayer region.
Using neutron scattering—a technique uniquely suited to probing light elements in aqueous media—the authors measured the d‑spacing of the vermiculite gel under isothermal conditions while incrementally adding aqueous solutions of either D‑histidine or L‑histidine at identical concentrations and pH. The key observation is that the two enantiomers produce opposite, statistically significant shifts in the basal spacing: D‑histidine contracts the interlayer distance by roughly 0.03 nm, whereas L‑histidine expands it by about 0.02 nm. The effect is reproducible across multiple runs and persists over a range of temperatures (10–30 °C) and ionic strengths, indicating that the response is intrinsic to the chiral interaction rather than an experimental artifact.
The authors discuss three mechanistic scenarios that could account for this chiral specificity. First, the protonation state of histidine’s imidazole ring varies with pH, altering its net charge distribution. Because the vermiculite interlayer contains a structured water network and exchangeable cations, the electrostatic field experienced by the layers is modulated differently by the two enantiomers. Second, the spatial arrangement of hydrogen‑bond donors and acceptors on the D‑ and L‑forms leads to distinct water‑mediated hydrogen‑bonding patterns, generating opposite mechanical stresses on the clay sheets. Third, the vermiculite surface itself is not perfectly symmetric; subtle variations in surface charge density and defect sites can preferentially stabilize one enantiomeric orientation over the other, lowering the free energy of that configuration. The paper acknowledges that distinguishing among these possibilities will require complementary spectroscopic probes (e.g., solid‑state NMR, X‑ray absorption spectroscopy) and atomistic simulations (MD, DFT) to map the charge distribution and hydrogen‑bond network at sub‑angstrom resolution.
From a prebiotic chemistry perspective, the findings are significant. Layered clays such as vermiculite were abundant on the early Earth and are known to adsorb and concentrate organic molecules from dilute aqueous solutions. If one enantiomer is preferentially retained or induces a subtle structural change in the clay lattice, repeated cycles of adsorption, desorption, and mineral transformation could amplify a small initial chiral bias. Histidine is particularly interesting because its imidazole side chain can coordinate transition‑metal ions (e.g., Ni²⁺, Cu²⁺), potentially linking chiral selection to metal‑catalyzed peptide bond formation or redox chemistry. Thus, the observed chiral shift in d‑spacing may represent a physical manifestation of a feedback loop where mineral structure and organic chirality co‑evolve.
The authors also place their work in the context of meteoritic organics. Recent analyses of the Murchison meteorite have shown an excess of one iso‑valine enantiomer correlated with the degree of aqueous alteration the meteorite experienced. By demonstrating that a simple clay mineral can distinguish between histidine enantiomers, the study provides a terrestrial analogue for how extraterrestrial organics could have been further processed on early Earth, potentially enhancing any pre‑existing enantiomeric excess delivered by meteorites.
Limitations of the study include the relatively narrow range of experimental conditions (single pH, limited temperature window) and the lack of direct structural data on how histidine molecules are oriented within the interlayer. The authors propose future work involving high‑resolution neutron reflectometry, isotope labeling, and computational modeling to quantify the binding geometry and energetics of each enantiomer.
In summary, the paper delivers the first quantitative, neutron‑scattering evidence that a hydrated, ordered vermiculite clay exhibits enantio‑selective swelling behavior in response to D‑ and L‑histidine. This result supports the hypothesis that mineral surfaces could have acted as chiral amplifiers in the prebiotic world, bridging the gap between the modest enantiomeric excesses observed in meteoritic organics and the near‑perfect homochirality of contemporary biomolecules.
Comments & Academic Discussion
Loading comments...
Leave a Comment