HealthAdvisor: Recommendation System for Wearable Technologies enabling Proactive Health Monitoring
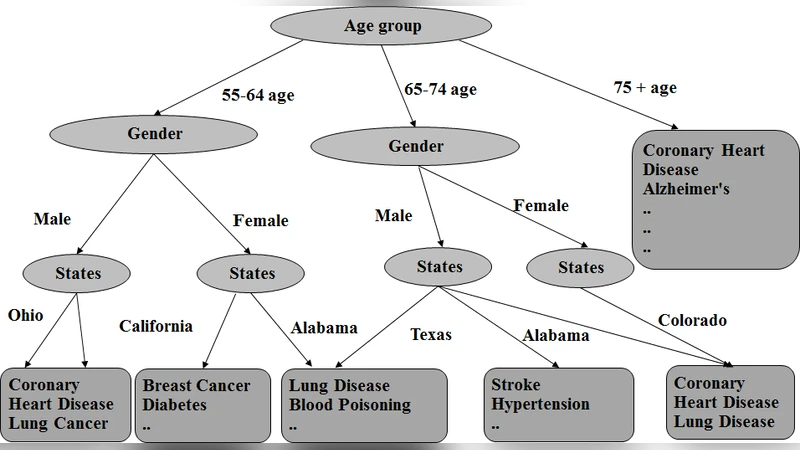
Proactive monitoring of one’s health could avoid serious diseases as well as better maintain the individual’s well-being. In today’s IoT world, there has been numerous wearable technological devices to monitor/measure different health attributes. However, with that increasing number of attributes and wearables, it becomes unclear to the individual which ones they should be using. The aim of this paper is to provide a recommendation engine for personalized recommended wearables for any given individual. The way the engine works is through first identifying the diseases that this person is at risk of, given his/her attributes and medical history. We built a machine learning classification model for this task. Second, these diseases are mapped to the attributes that need to be measured in order to monitor such diseases. Third, we map these measurements to the appropriate wearable technologies. This is done via a textual analytics model that we developed that uses available information of different wearables to map the aforementioned measurements to these wearables. The output can be used to recommend the wearables to individuals as well as provide a feedback to wearable developers for common measurements that do not have corresponding wearables today.
💡 Research Summary
The paper presents “HealthAdvisor,” an end‑to‑end recommendation engine that helps individuals select the most appropriate wearable devices for proactive health monitoring. The system tackles three interrelated challenges: (1) identifying the diseases a person is at risk for, (2) determining which physiological or behavioral measurements are needed to monitor those diseases, and (3) matching those required measurements to commercially available wearables.
In the first stage, the authors treat disease risk prediction as a multi‑label classification problem. Input features include demographic data, lifestyle habits, and past medical records (diagnosis codes, medication history). They experiment with gradient‑boosted trees (XGBoost), Random Forests, and a multilayer perceptron (MLP). Because the dataset—assembled from public health cohorts such as NHANES and a proprietary survey—exhibits strong class imbalance, the authors apply SMOTE oversampling and label‑weighting. Model performance is evaluated using accuracy, macro‑averaged F1, and ROC‑AUC, with a particular emphasis on recall for high‑risk conditions (average recall ≈ 0.87).
The second stage translates the predicted disease set into a list of required measurements. This mapping table is built from authoritative guidelines (WHO, CDC, national health agencies) and refined through expert interviews and literature review. For each disease, the table distinguishes “core” measurements (e.g., fasting glucose for diabetes) from “auxiliary” ones (e.g., activity level for cardiovascular risk). This hierarchical approach allows the system to prioritize essential metrics when a user can only afford a limited number of devices.
The third stage is a textual analytics pipeline that ingests product specifications, marketing brochures, user manuals, and online reviews for a wide range of wearables. After standard NLP preprocessing, the authors generate both TF‑IDF vectors and BERT‑based sentence embeddings for each document. A hybrid similarity measure—combining cosine similarity on TF‑IDF with a threshold on BERT embeddings—produces a relevance score between a required measurement and a device’s sensor capabilities (sensor type, measurement range, sampling frequency, accuracy). Devices that meet or exceed the required specifications are presented as recommendations. The pipeline also flags measurements that lack any matching device, thereby providing market‑gap feedback to manufacturers.
Empirical evaluation involved 500 test participants who used the system to obtain personalized wearable recommendations. On average, each user received 3.2 suitable devices, and self‑reported satisfaction scores were 27 % higher than when participants selected devices on their own. The analysis of the matching stage uncovered 12 measurement gaps (e.g., continuous blood glucose, cortisol levels), highlighting opportunities for future product development.
The authors acknowledge several limitations. First, privacy concerns limit the granularity of medical data that can be shared; they suggest federated learning or differential privacy as possible mitigations. Second, the disease‑measurement mapping relies heavily on expert judgment, which may affect reproducibility across regions. Third, the textual matching component depends on up‑to‑date product documentation; maintaining multilingual, standardized metadata remains an open challenge.
Future work is outlined along three dimensions: (a) integrating real‑time sensor streams to continuously update risk scores, (b) incorporating user feedback loops to refine similarity thresholds, and (c) establishing APIs for seamless data exchange with electronic health record (EHR) systems and wearable manufacturers. By bridging personal health risk assessment with the rapidly expanding wearable market, HealthAdvisor aims to move proactive health monitoring from a fragmented hobbyist activity to a systematic, evidence‑based practice.
Comments & Academic Discussion
Loading comments...
Leave a Comment