Multi-Constituent Simulation of Thrombus Deposition
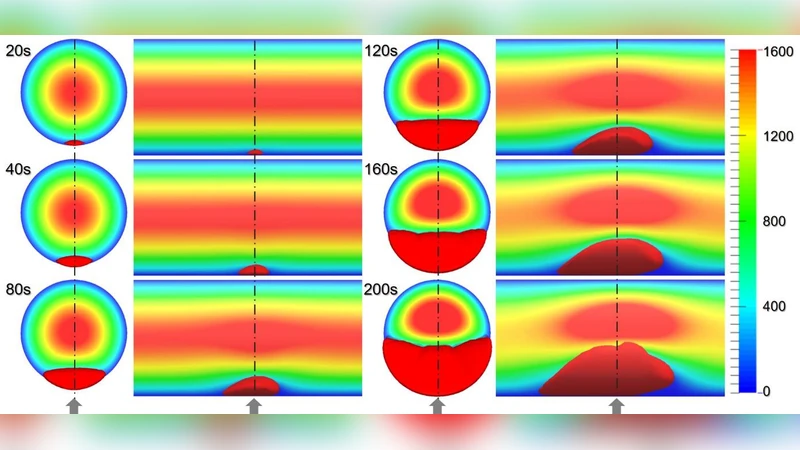
In this paper, we present a spatio-temporal mathematical model for simulating the formation and growth of a thrombus. Blood is treated as a multi-constituent mixture comprised of a linear fluid phase and a thrombus (solid) phase. The transport and reactions of 10 chemical and biological species are incorporated using a system of coupled convection-reaction-diffusion (CRD) equations to represent three processes in thrombus formation: initiation, propagation and stabilization. Computational fluid dynamic (CFD) simulations using the libraries of OpenFOAM were performed for two illustrative benchmark problems: in vivo thrombus growth in an injured blood vessel and in vitro thrombus deposition in micro-channels (1.5mm x 1.6mm x 0.1mm) with small crevices (125{\mu}m x 75{\mu}m and 125{\mu}m x 137{\mu}m). For both problems, the simulated thrombus deposition agreed very well with experimental observations, both spatially and temporally. Based on the success with these two benchmark problems, which have very different flow conditions and biological environments, we believe that the current model will provide useful insight into the genesis of thrombosis in blood-wetted devices, and provide a tool for the design of less thrombogenic devices.
💡 Research Summary
The paper introduces a comprehensive spatio‑temporal model that treats blood as a two‑phase mixture consisting of a linear fluid phase and a solid thrombus phase. Ten biochemical species—representing key mediators such as ADP, thromboxane A2, thrombin, fibrin precursors, and inhibitory proteins—are governed by coupled convection‑reaction‑diffusion (CRD) equations. The reaction terms combine first‑order kinetics with Michaelis‑Menten formulations, allowing the model to capture initiation (platelet activation), propagation (thrombin‑driven fibrin formation), and stabilization (cross‑linking and inhibition) processes within a single framework.
The governing equations for the fluid phase are the incompressible Navier‑Stokes equations, while the thrombus phase is represented by a scalar volume‑fraction field φ that evolves according to a transport equation containing source and sink terms linked to the biochemical reactions. This approach avoids the computational overhead of full solid mechanics while still providing a physically meaningful measure of thrombus growth.
Implementation is carried out in OpenFOAM. Temporal discretization uses a second‑order backward‑difference (BDF2) scheme; spatial discretization employs second‑order central differencing with a Total Variation Diminishing (TVD) limiter to reduce numerical diffusion. Boundary conditions are matched to experimental setups: prescribed inlet velocity profiles, zero‑gradient outlet pressure, and no‑slip, no‑flux walls. Mesh refinement is applied around geometric features such as crevices to resolve local recirculation zones accurately.
Two benchmark problems validate the model. The first simulates in‑vivo thrombus formation in an injured arterial segment. The model reproduces the rapid increase in thrombus volume observed experimentally, showing that the localized drop in shear stress and the ensuing platelet activation are captured correctly. The second benchmark reproduces in‑vitro thrombus deposition in micro‑channels (1.5 mm × 1.6 mm × 0.1 mm) containing two rectangular crevices (125 µm × 75 µm and 125 µm × 137 µm). Simulations predict that low‑velocity recirculation within the crevices leads to early platelet accumulation and subsequent fibrin polymerization, matching the spatial pattern and timing of thrombus growth measured in laboratory experiments.
The authors discuss strengths such as the unified treatment of fluid dynamics, chemistry, and solid deposition; the ability to handle multiple interacting species; and the open‑source, reproducible nature of the OpenFOAM implementation. Limitations include the simplified mechanical description of the thrombus (no explicit elasticity or fracture), reliance on experimentally calibrated kinetic parameters, and the high computational cost associated with fine meshes required for crevice‑scale resolution.
In conclusion, the presented multi‑constituent framework successfully predicts thrombus formation under diverse flow conditions and biological environments, offering a valuable tool for evaluating thrombogenic risk in blood‑wetted medical devices. Future work is suggested to incorporate nonlinear solid mechanics for mature thrombus, patient‑specific parameter identification, and multiscale coupling to bridge microscale platelet dynamics with organ‑level hemodynamics.
Comments & Academic Discussion
Loading comments...
Leave a Comment