Adverse Events in Robotic Surgery: A Retrospective Study of 14 Years of FDA Data
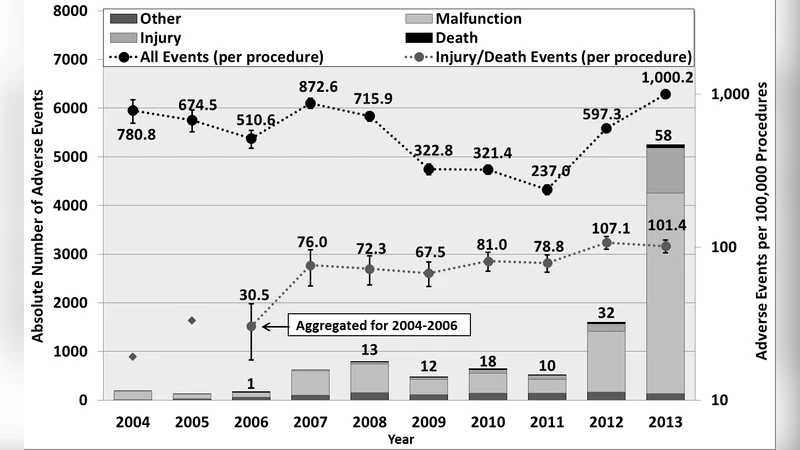
Understanding the causes and patient impacts of surgical adverse events will help improve systems and operational practices to avoid incidents in the future. We analyzed the adverse events data related to robotic systems and instruments used in minimally invasive surgery, reported to the U.S. FDA MAUDE database from January 2000 to December 2013. We determined the number of events reported per procedure and per surgical specialty, the most common types of device malfunctions and their impact on patients, and the causes for catastrophic events such as major complications, patient injuries, and deaths. During the study period, 144 deaths (1.4% of the 10,624 reports), 1,391 patient injuries (13.1%), and 8,061 device malfunctions (75.9%) were reported. The numbers of injury and death events per procedure have stayed relatively constant since 2007 (mean = 83.4, 95% CI, 74.2-92.7). Surgical specialties, for which robots are extensively used, such as gynecology and urology, had lower number of injuries, deaths, and conversions per procedure than more complex surgeries, such as cardiothoracic and head and neck (106.3 vs. 232.9, Risk Ratio = 2.2, 95% CI, 1.9-2.6). Device and instrument malfunctions, such as falling of burnt/broken pieces of instruments into the patient (14.7%), electrical arcing of instruments (10.5%), unintended operation of instruments (8.6%), system errors (5%), and video/imaging problems (2.6%), constituted a major part of the reports. Device malfunctions impacted patients in terms of injuries or procedure interruptions. In 1,104 (10.4%) of the events, the procedure was interrupted to restart the system (3.1%), to convert the procedure to non-robotic techniques (7.3%), or to reschedule it to a later time (2.5%). Adoption of advanced techniques in design and operation of robotic surgical systems may reduce these preventable incidents in the future.
💡 Research Summary
This retrospective analysis examined adverse events associated with robotic-assisted minimally invasive surgery reported to the U.S. Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database over a fourteen‑year period (January 2000–December 2013). A total of 10,624 reports were identified, comprising 144 deaths (1.4 % of reports), 1,391 patient injuries (13.1 %), and 8,061 device malfunctions (75.9 %). The annual rates of injury and death per procedure stabilized after 2007, with a mean of 83.4 events per year (95 % CI = 74.2–92.7), indicating that the early learning curve did not translate into a sustained reduction in severe outcomes.
When stratified by surgical specialty, procedures in gynecology and urology—areas with the highest robotic utilization—experienced substantially fewer adverse events per case than more technically demanding specialties such as cardiothoracic and head‑and‑neck surgery. The injury/death rate was 106.3 per 1,000 procedures in gynecology/urology versus 232.9 in cardiothoracic/head‑and‑neck, yielding a risk ratio of 2.2 (95 % CI = 1.9–2.6). This suggests that procedural complexity and anatomical constraints amplify the clinical impact of device failures.
Device‑related malfunctions dominated the adverse event landscape. The most frequent failure mode was the loss of burnt or broken instrument fragments into the patient (14.7 % of device reports), followed by electrical arcing (10.5 %), unintended instrument activation (8.6 %), system‑wide software or hardware errors (5.0 %), and video/imaging problems (2.6 %). These malfunctions directly contributed to patient injury and indirectly caused procedural interruptions. In 1,104 cases (10.4 % of all reports), the surgery was halted: 3.1 % required a system restart, 7.3 % were converted to conventional (non‑robotic) techniques, and 2.5 % were postponed or rescheduled. Such interruptions increase operative time, anesthesia exposure, and overall healthcare costs.
Methodologically, the study extracted all relevant MAUDE entries, removed duplicates through cross‑referencing of event identifiers and dates, and classified each report according to a predefined taxonomy (death, injury, malfunction type). Trend analysis employed moving averages and 95 % confidence intervals, while specialty‑specific risk ratios were derived from logistic regression models controlling for procedure volume. The authors acknowledge inherent limitations of the MAUDE system: voluntary reporting leads to under‑reporting, manufacturer submissions may be delayed, and inconsistent terminology hampers uniform classification. Consequently, absolute incidence rates may be underestimated, but relative comparisons remain informative.
The authors conclude that, although robotic surgery has an overall favorable safety profile, device malfunctions represent the primary source of adverse events and are disproportionately consequential in high‑complexity specialties. They advocate for engineering improvements—such as redundant safety checks, real‑time fault detection, and more intuitive user interfaces—to mitigate preventable failures. Additionally, institutional measures like routine system diagnostics, standardized emergency protocols, and comprehensive staff training are recommended. Future research should integrate intra‑operative log data and electronic health records to achieve more granular causality assessments and to evaluate the impact of newer robotic platforms on patient safety.