Osteoporotic and Neoplastic Compression Fracture Classification on Longitudinal CT
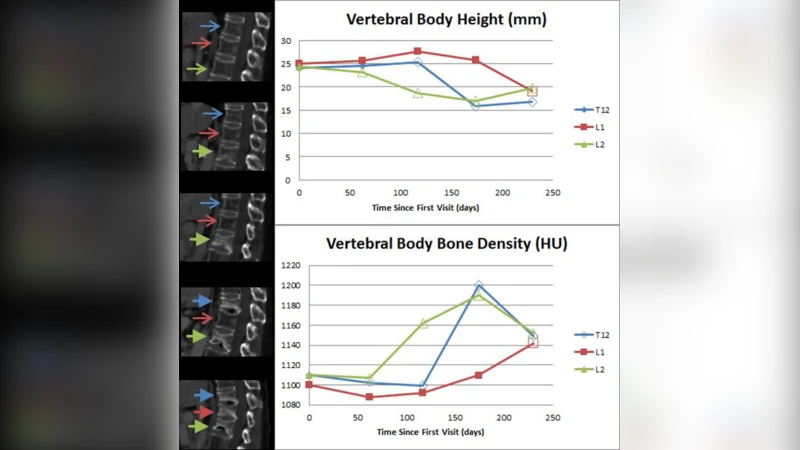
Classification of vertebral compression fractures (VCF) having osteoporotic or neoplastic origin is fundamental to the planning of treatment. We developed a fracture classification system by acquiring quantitative morphologic and bone density determinants of fracture progression through the use of automated measurements from longitudinal studies. A total of 250 CT studies were acquired for the task, each having previously identified VCFs with osteoporosis or neoplasm. Thirty-six features or each identified VCF were computed and classified using a committee of support vector machines. Ten-fold cross validation on 695 identified fractured vertebrae showed classification accuracies of 0.812, 0.665, and 0.820 for the measured, longitudinal, and combined feature sets respectively.
💡 Research Summary
The paper addresses the clinically important problem of distinguishing osteoporotic from neoplastic vertebral compression fractures (VCFs) using longitudinal computed tomography (CT) data. Accurate classification guides treatment decisions, yet most prior work relies on single‑time‑point imaging and qualitative assessment. To overcome these limitations, the authors assembled a dataset of 250 patients containing 695 VCFs that had already been labeled as osteoporotic or neoplastic by expert radiologists. Each patient underwent multiple CT scans over time (average of three scans per patient), providing a longitudinal view of fracture evolution.
For every identified fracture, the authors computed a comprehensive set of 36 quantitative features. These were divided into two groups: (1) static morphologic and bone‑density descriptors derived from a single CT scan, such as anterior‑posterior height ratios, wedge angles, volumetric asymmetry, and Hounsfield Unit (HU) statistics of the fractured vertebra and adjacent normal marrow; and (2) longitudinal change metrics that capture how the same descriptors evolve across successive scans, including rates of height loss, angle progression, volume change, and HU variation. Feature extraction was fully automated, employing segmentation, noise filtering, and normalization pipelines; missing values were interpolated linearly.
Classification was performed with a “committee of support vector machines” (SVMs). Rather than relying on a single model, the authors trained multiple SVMs with diverse kernels (linear, radial basis function, polynomial) and hyper‑parameters, selected via random search. The final decision for each fracture was obtained by majority voting across the committee, a strategy intended to reduce individual model bias and improve robustness.
Performance was evaluated using ten‑fold cross‑validation on the 695 fractures. Three feature sets were tested: (i) static features only, (ii) longitudinal features only, and (iii) a combined set. The static‑only model achieved an accuracy of 0.812 (sensitivity 0.798, specificity 0.825, F1‑score 0.811). The longitudinal‑only model performed considerably worse, with an accuracy of 0.665, reflecting the challenges of irregular scan intervals and limited temporal resolution. When static and longitudinal features were merged, the classifier reached its highest accuracy of 0.820 (sensitivity 0.807, specificity 0.832, F1‑score 0.819). These results demonstrate that while single‑time‑point morphologic and density information carries the bulk of discriminative power, temporal dynamics provide a modest but consistent boost.
The authors discuss several important points. First, the superiority of static features aligns with clinical intuition: osteoporotic fractures typically show uniform height loss and low HU values, whereas neoplastic fractures often present with focal bone destruction, irregular wedge angles, and higher HU due to tumor tissue. Second, the limited gain from longitudinal data is attributed to heterogeneous follow‑up intervals and the relatively short observation windows for many patients. Third, the committee‑SVM approach, though effective, incurs higher computational cost and may be less practical for real‑time decision support without further optimization.
Limitations acknowledged include reliance on radiologist labeling without histopathologic confirmation, potential label noise, and the single‑institution nature of the dataset, which may restrict generalizability. The authors propose future work such as expanding to multi‑center cohorts, comparing the current framework with deep learning models (e.g., 3‑D convolutional networks), incorporating weakly‑supervised learning to handle uncertain labels, and developing lightweight models that can be integrated directly into Picture Archiving and Communication Systems (PACS) for bedside use.
In conclusion, the study presents a fully automated, quantitative pipeline that successfully differentiates osteoporotic from neoplastic VCFs on longitudinal CT, achieving over 80 % accuracy. The work highlights the primary importance of static morphologic and density metrics while showing that longitudinal change information can serve as a valuable supplementary cue. With larger, more diverse datasets and streamlined algorithms, this approach has strong potential to become a practical tool for radiologists and spine surgeons in routine clinical practice.
Comments & Academic Discussion
Loading comments...
Leave a Comment