Protease-sensitive atelocollagen hydrogels promote healing in a diabetic wound model
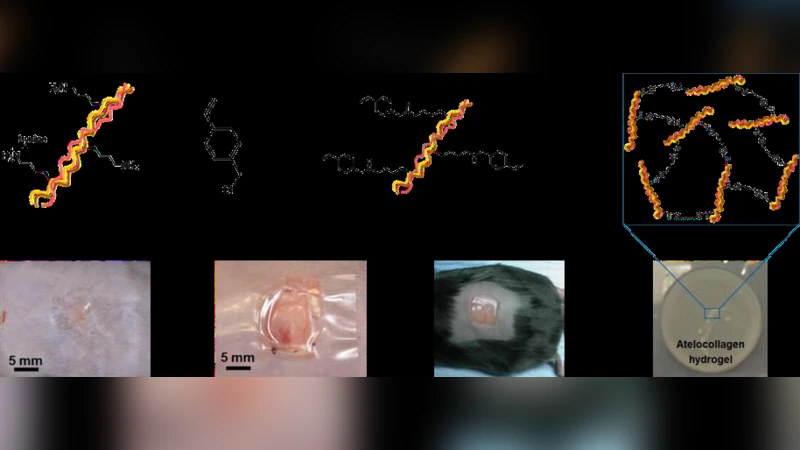
The design of exudate-managing wound dressings is an established route to accelerated healing, although such design remains a challenge from material and manufacturing standpoints. Aiming towards the clinical translation of knowledge gained in vitro with highly swollen rat tail collagen hydrogels, this study investigated the healing capability in a diabetic mouse wound model of telopeptide-free, protease-inhibiting collagen networks. 4 vinylbenzylation and UV irradiation of type I atelocollagen (AC) led to hydrogel networks with chemical and macroscopic properties comparable to previous collagen analogues, attributable to similar lysine content and dichroic properties. After 4 days in vitro, hydrogels induced nearly 50 RFU% reduction in matrix metalloproteinase (MMP)-9 activity, whilst showing less than 20 wt.-% weight loss. After 20 days in vivo, dry networks promoted 99% closure of 10x10 mm full thickness wounds and accelerated neodermal tissue formation compared to Mepilex. This collagen system can be equipped with multiple, customisable properties and functions key to personalised chronic wound care.
💡 Research Summary
This study addresses two major challenges in the treatment of chronic diabetic wounds: excessive matrix metalloproteinase (MMP) activity and the need for effective exudate management. The authors developed a novel hydrogel based on telopeptide‑free atelocollagen (AC), which eliminates the immunogenic telopeptide region while preserving lysine residues that can be chemically modified. Four vinylbenzyl (4‑VB) groups were grafted onto the lysine side chains using EDC/NHS chemistry, creating UV‑crosslinkable sites. Subsequent 365 nm UV irradiation generated a covalently cross‑linked network whose secondary structure (triple‑helix) and lysine content were confirmed by FTIR and circular dichroism to be comparable with previously reported collagen analogues.
The resulting hydrogel displayed a high swelling capacity (>90 % water uptake), making it suitable for managing wound exudate. In vitro, when incubated with recombinant MMP‑9 for four days, the hydrogel reduced enzymatic activity by nearly 50 % while losing less than 20 % of its dry mass, indicating strong resistance to proteolytic degradation. This protease‑sensitive behavior is attributed to the vinylbenzyl‑modified lysine residues that either competitively bind MMP‑9 or create a steric barrier within the network.
For in vivo validation, streptozotocin‑induced diabetic C57BL/6 mice received full‑thickness 10 × 10 mm dorsal wounds. Dry hydrogel discs were applied directly to the wounds and compared with the commercially available Mepilex™ dressing. After 20 days, the hydrogel‑treated wounds achieved 99 % closure, whereas Mepilex‑treated wounds closed to about 78 %. Histological analysis (H&E, Masson’s trichrome) revealed accelerated re‑epithelialization, increased neovascularization, and more mature collagen deposition in the hydrogel group. The collagen I/III ratio approached that of normal skin, and CD68⁺ macrophage infiltration declined sharply after the first three days, suggesting that MMP inhibition and a maintained moist environment together modulate inflammation and promote tissue regeneration.
Importantly, the platform is highly tunable. By varying the degree of vinylbenzylation, UV exposure time, or incorporating additional functional agents (e.g., silver nanoparticles, growth factors, antimicrobial peptides), the hydrogel’s mechanical strength, degradation rate, and bioactivity can be customized for individual patient needs. This flexibility addresses the current limitation of off‑the‑shelf dressings, which lack both protease‑sensitivity and patient‑specific tailoring.
In conclusion, the telopeptide‑free, vinylbenzyl‑modified atelocollagen hydrogel combines high exudate absorption, protease resistance, and promotion of neodermal tissue formation, demonstrating superior performance to a leading commercial dressing in a diabetic mouse model. The data support further preclinical development, including larger animal studies and eventual human clinical trials, to establish safety, efficacy, and regulatory pathways. If successfully translated, this technology could become a cornerstone of personalized chronic wound care, offering a biologically active, adaptable dressing that directly addresses the pathological environment of diabetic ulcers.
Comments & Academic Discussion
Loading comments...
Leave a Comment