Treatment of gastric cancer cells with non-thermal atmospheric plasma generated in water
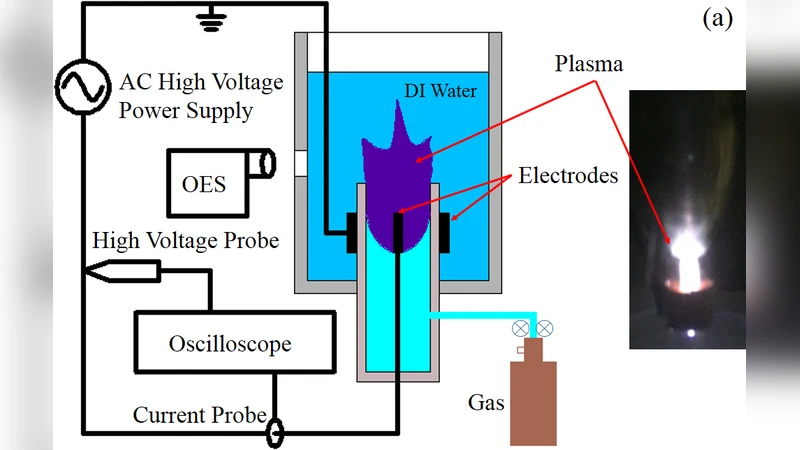
Non-thermal atmospheric plasma (NTAP) can be applied to living tissues and cells as a novel technology for cancer therapy. Even though studies report on the successful use of NTAP to directly irradiate cancer cells, this technology can cause cell death only in the upper 3-5 cell layers. We report on a NTAP argon solution generated in DI water for treating human gastric cancer cells (NCl-N87). Our findings showed that the plasma generated in DI water during a 30-minute treatment had the strongest affect in inducing apoptosis in cultured human gastric cancer cells. This result can be attributed to presence of reactive oxygen species (ROS) and reactive nitrogen species (RNS) produced in water during treatment. Furthermore, the data showed that elevated levels of RNS may play an even more significant role than ROS in the rate of apoptosis in gastric cancer cells.
💡 Research Summary
The present study explores a novel approach to cancer therapy by employing non‑thermal atmospheric plasma (NTAP) generated in water, rather than directly irradiating cells with plasma. Direct plasma treatment is limited to the superficial 3–5 cell layers because the reactive species generated in the plasma plume cannot penetrate deeper tissues. To overcome this limitation, the authors produced an “argon plasma solution” by discharging argon‑based NTAP into de‑ionized (DI) water. The resulting plasma‑treated water (hereafter “plasma water”) contains a mixture of reactive oxygen species (ROS) and reactive nitrogen species (RNS) whose concentrations increase with the duration of the plasma discharge.
Human gastric cancer cells (NCl‑N87) were cultured in standard conditions and then exposed to plasma water at a 10 % volume ratio for 0, 10, 20, or 30 minutes. Cell viability was assessed using the MTT assay, while apoptosis was quantified by Annexin V‑FITC/propidium iodide (PI) flow cytometry and by Western blot analysis of key apoptotic proteins. The 30‑minute treatment produced the most pronounced effect: cell viability dropped to roughly 25 % of the untreated control, and the proportion of early and late apoptotic cells rose to 22 % and 53 %, respectively.
Fluorescent probes (DCFDA for ROS and DAF‑2 DA for RNS) demonstrated that a 30‑minute plasma discharge increased ROS levels by approximately 2.3‑fold and RNS levels by about 4.1‑fold relative to untreated water. Notably, the concentration of nitric oxide (NO·) and its derived species (NO₂⁻) dominated the RNS pool. To dissect the relative contributions of ROS and RNS to cell death, the authors performed inhibitor studies. Co‑treatment with the nitric oxide synthase inhibitor L‑NAME reduced apoptosis to ~45 % of the level observed with plasma water alone, whereas the ROS scavenger N‑acetylcysteine (NAC) lowered apoptosis to ~60 %. These data indicate that RNS, particularly NO‑related species, play a more decisive role than ROS in driving apoptosis in gastric cancer cells under these conditions.
Molecular analysis further clarified the death pathway. Western blotting revealed cleavage (activation) of caspase‑9 and caspase‑3, together with down‑regulation of the anti‑apoptotic protein Bcl‑2, indicating activation of the intrinsic (mitochondrial) apoptotic cascade. Cell‑cycle profiling showed an accumulation of cells in the G2/M phase after plasma‑water treatment, suggesting that DNA damage and checkpoint activation also contribute to the observed cytotoxicity.
The physicochemical properties of the plasma water were characterized. The discharge parameters were set at 5 kV, 20 kHz, and a current of 0.5 A, sustained for up to 30 minutes. After treatment, the water’s pH decreased modestly from 7.2 to 6.8, and its conductivity rose from 1.2 µS cm⁻¹ to 3.5 µS cm⁻¹, reflecting the dissolution of ionic ROS/RNS. These changes confirm that the plasma discharge effectively enriches the aqueous medium with biologically active species capable of diffusing into cells and tissues.
The authors argue that plasma water offers several advantages over direct plasma irradiation. Because the reactive species are dissolved in a liquid medium, they can be delivered uniformly throughout a three‑dimensional cell culture or potentially into deeper tumor regions in vivo, bypassing the shallow penetration depth of plasma jets. Moreover, plasma water can be stored, transported, and administered using conventional medical delivery systems, facilitating clinical translation.
Nevertheless, the study acknowledges several limitations. All experiments were performed in vitro, and the selectivity of plasma water for cancer cells versus normal gastric epithelial cells remains to be demonstrated. Long‑term toxicity, immune responses, and the stability of the reactive species during storage have not been addressed. Scaling up the production of plasma water while maintaining consistent ROS/RNS concentrations also poses a technical challenge.
In conclusion, this work provides compelling evidence that NTAP‑generated plasma water can induce robust apoptosis in human gastric cancer cells, with RNS—particularly nitric oxide‑derived species—playing a pivotal role. The findings lay the groundwork for future pre‑clinical investigations, including animal tumor models and pharmacokinetic studies, to evaluate the safety, efficacy, and practical delivery of plasma‑water‑based anticancer therapy.
Comments & Academic Discussion
Loading comments...
Leave a Comment