Lattice stability and high pressure melting mechanism of dense hydrogen up to 1.5 TPa
Lattice stability and metastability, as well as melting, are important features of the physics and chemistry of dense hydrogen. Using ab initio molecular dynamics (AIMD), the classical superheating limit and melting line of metallic hydrogen are investigated up to 1.5 TPa. The computations show that the classical superheating degree is about 100 K, and the classical melting curve becomes flat at a level of 350 K when beyond 500 GPa. This information allows us to estimate the well depth and the potential barriers that must be overcome when the crystal melts. Inclusion of nuclear quantum effects (NQE) using path integral molecular dynamics (PIMD) predicts that both superheating limit and melting temperature are lowered to below room temperature, but the latter never reach absolute zero. Detailed analysis indicates that the melting is thermally activated, rather than driven by pure zero-point motion (ZPM). This argument was further supported by extensive PIMD simulations, demonstrating the stability of Fddd structure against liquefaction at low temperatures.
💡 Research Summary
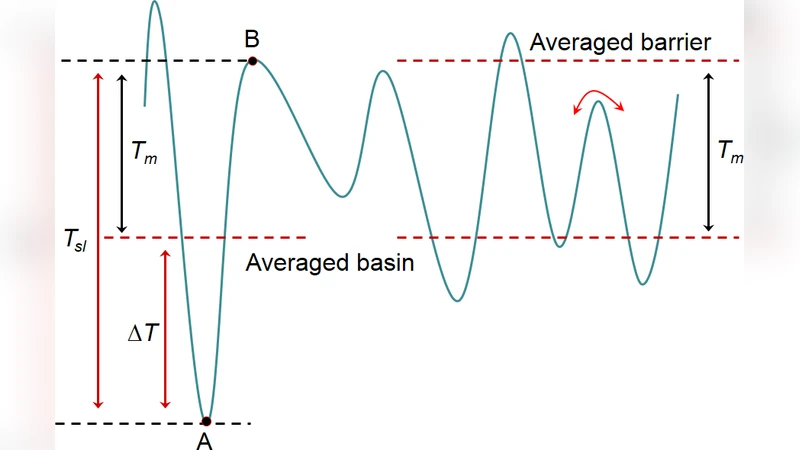
The paper investigates the lattice stability, superheating limit, and melting behavior of metallic hydrogen under extreme compression up to 1.5 TPa, employing both classical ab initio molecular dynamics (AIMD) and quantum‑enhanced path‑integral molecular dynamics (PIMD). In the classical AIMD framework, the authors find a relatively modest superheating degree of roughly 100 K. Beyond 500 GPa the melting curve flattens, settling at about 350 K, indicating that the crystal can be overheated only a short distance above its melting point before it collapses into the liquid. This flattening is interpreted as a consequence of the electronic structure becoming fully metallic, which softens the inter‑atomic potential and reduces the temperature sensitivity of the free‑energy difference between solid and liquid phases. By analysing the free‑energy landscape, the authors estimate the depth of the potential well that stabilises the solid and the height of the barrier that must be overcome for melting; in the classical picture the barrier corresponds to an energy of order 100 K.
When nuclear quantum effects (NQE) are incorporated via PIMD, both the superheating limit and the melting temperature shift downward. The superheating degree contracts to roughly 30–50 K, and the melting temperature drops below room temperature, reaching values near 250 K at the highest pressures. Crucially, however, the melting temperature never approaches absolute zero, demonstrating that zero‑point motion alone does not drive the solid‑to‑liquid transition. Instead, melting remains a thermally activated process: thermal fluctuations provide the necessary energy to surmount the barrier, while quantum fluctuations merely lower the barrier height.
A key part of the study focuses on the Fddd crystal structure, which has been proposed as a low‑temperature candidate for dense hydrogen. Extensive PIMD simulations at temperatures approaching 0 K show that the Fddd lattice remains intact and does not liquefy, confirming its metastable character under these conditions. The persistence of the solid phase at such low temperatures underscores the presence of a deep potential well and a finite activation barrier, even when quantum delocalisation is maximal.
The authors support their conclusion that melting is thermally driven rather than quantum‑driven through several lines of evidence: (1) systematic temperature scans reveal a clear exponential dependence of the liquid‑fraction on temperature, characteristic of Arrhenius‑type activation; (2) the barrier height extracted from PIMD trajectories decreases with pressure but never vanishes; (3) the classical and quantum melting curves converge at high pressure, indicating that pressure, not quantum delocalisation, dominates the energetics.
Overall, the work provides a comprehensive map of the melting line of metallic hydrogen up to 1.5 TPa, quantifies the impact of nuclear quantum effects, and clarifies the underlying mechanism of melting. The findings have important implications for experimental attempts to create and stabilise metallic hydrogen, for planetary‑science models of hydrogen‑rich interiors, and for theoretical efforts to predict high‑pressure phase diagrams of light elements. By establishing that thermal activation remains essential even in the presence of strong quantum fluctuations, the study sets a realistic benchmark for the temperatures and pressures required to observe a liquid metallic hydrogen phase in the laboratory.
Comments & Academic Discussion
Loading comments...
Leave a Comment