A Computational Model of YAP/TAZ Mechanosensing
In cell proliferation, stem cell differentiation, chemoresistance and tissue organization, the ubiquitous role of YAP/TAZ continues to impact our fundamental understanding in numerous physiological and disease systems. YAP/TAZ is an important signaling nexus integrating diverse mechanical and biochemical signals, such as ECM stiffness, adhesion ligand density, or cell-cell contacts, and thus strongly influences cell fate. Recent studies show that YAP/TAZ mechanical sensing is dependent on RhoA-regulated stress fibers. However, current understanding of YAP/TAZ still remains limited due to the unknown interaction between the canonical Hippo pathway and cell tension. To identify the roles of key signaling molecules in mechanical signal sensing and transduction, we present a novel computational model of the YAP/TAZ signaling pathway. This model converts ECM mechanical properties to biochemical signals via adhesion, and integrates intracellular signaling cascades associated with cytoskeleton dynamics. Adhesion molecules, such as FAK, are predicted to rescue YAP/TAZ activity in soft environments via the RhoA pathway. We found that changes of molecule concentrations result in different pattern of YAP/TAZ stiffness response. We also investigate the sensitivity of YAP/TAZ activity to ECM stiffness. In addition, the model shows that the unresolved synergistic effect of YAP/TAZ activity between the mechanosensing and the Hippo pathways can be explained by the interaction of LIMK and LATS. Overall, our model provides a novel platform for studying YAP/TAZ activity in the context of integrating different signaling pathways. This platform can be used to gain new fundamental insights into roles of key molecular and mechanical regulators on development, tissue engineering or tumor progression.
💡 Research Summary
The manuscript presents a systems‑level computational model that links extracellular matrix (ECM) mechanics to the biochemical activity of the YAP/TAZ transcriptional co‑activators. Recognizing that YAP/TAZ integrates both canonical Hippo signaling and mechanical cues such as substrate stiffness, ligand density, and cell‑cell contacts, the authors construct a set of ordinary differential equations that capture three interconnected modules: (1) mechanotransduction at focal adhesions, where ECM stiffness and ligand presentation drive activation of focal adhesion kinase (FAK); (2) cytoskeletal dynamics, in which FAK‑stimulated RhoA‑GTP activates ROCK and mDia, leading to actin stress‑fiber formation and LIM kinase (LIMK)‑mediated cofilin phosphorylation; and (3) Hippo pathway modulation, where the assembled stress fibers inhibit LATS1/2, thereby reducing YAP/TAZ phosphorylation and permitting nuclear translocation. Parameter values are drawn from published kinetic data and calibrated against published experimental observations.
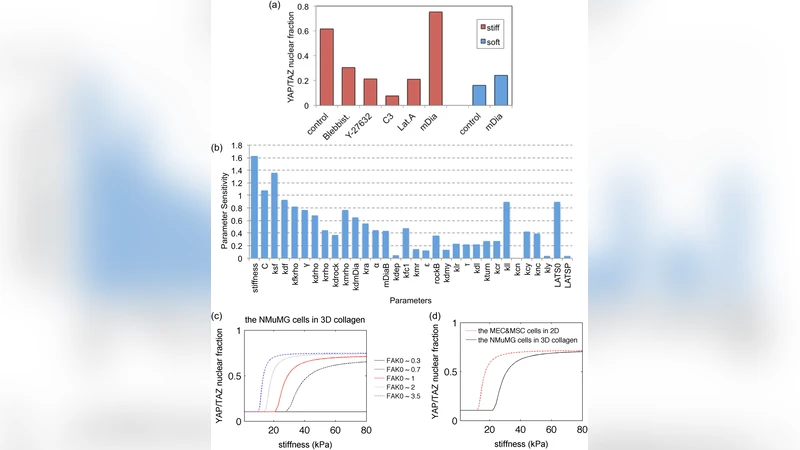
Simulation results reveal several key insights. First, the model predicts a “mechanical rescue” phenomenon: even on soft substrates, overexpression or pharmacological activation of FAK restores RhoA‑ROCK‑LIMK signaling, stabilizes stress fibers, suppresses LATS activity, and consequently re‑activates nuclear YAP/TAZ. This aligns with experimental reports that FAK can compensate for low matrix rigidity. Second, systematic variation of initial protein concentrations shows that the YAP/TAZ response to stiffness is not a simple sigmoidal curve but exhibits multiple switch‑like regimes. For instance, high LATS levels blunt stiffness sensitivity, whereas increasing LIMK can re‑establish a steep response. Third, the long‑standing observation of a synergistic effect between Hippo and mechanosensing pathways is explained by a direct or indirect interaction between LIMK and LATS: LIMK either binds LATS or, via ROCK‑mediated phosphorylation, reduces LATS kinase activity, thereby coupling cytoskeletal tension to Hippo output.
Global sensitivity analysis identifies ECM stiffness, FAK activation rate, and the fraction of RhoA‑GTP as the most influential parameters governing nuclear YAP/TAZ levels. LIMK‑cofilin dynamics emerge as a critical intermediary that translates stress‑fiber stability into Hippo modulation. Parameter space exploration uncovers a “stiffness‑independent” regime (high FAK, low LATS, moderate LIMK) where YAP/TAZ remains active regardless of substrate rigidity, offering a plausible mechanistic explanation for cancer cells that sustain proliferative signaling in soft microenvironments.
The authors validate the model by reproducing experimental perturbations: FAK inhibition diminishes YAP/TAZ nuclear localization, while LIMK activation rescues it even when LATS is abundant. These concordances demonstrate that the model captures essential biological behavior and can be used predictively.
In conclusion, this work delivers the first integrated computational framework that quantitatively links ECM mechanics, focal adhesion signaling, cytoskeletal tension, and Hippo pathway activity to YAP/TAZ output. By elucidating how LIMK and LATS mediate cross‑talk, the model provides mechanistic clarity for previously ambiguous synergistic effects. The platform is poised to guide experimental design, identify therapeutic targets (e.g., LIMK or FAK modulators), and inform tissue‑engineering strategies where precise control of YAP/TAZ activity is desired.
Comments & Academic Discussion
Loading comments...
Leave a Comment