Extracellular stimulation of nerve cells with electric current spikes induced by voltage steps
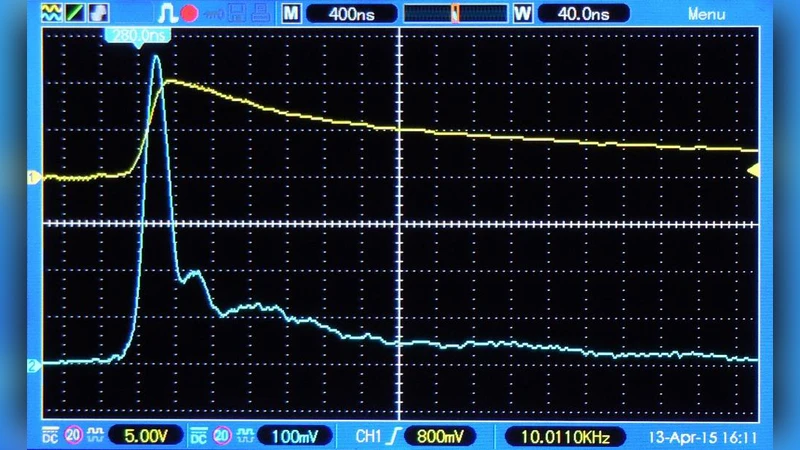
A new stimulation paradigm is presented for the stimulation of nerve cells by extracellular electric currents. In the new paradigm stimulation is achieved with the current spike induced by a voltage step whenever the voltage step is applied to a live biological tissue. By experimental evidence and theoretical arguments, it is shown that this spike is well suited for the stimulation of nerve cells. Stimulation of the human tongue is used for proof of principle. Charge injection thresholds are measured for various voltages. The time-profile of the current spike used in the experiment has a half-width of about 1 microsecond. The decay of the spike is non-exponential. The spike has at least three distinctly different phases. A Maxwell phase is followed by a charge-rearrangement phase. Charging of cell membranes is completed in a third phase. All three phases contribute to depolarization or hyperpolarization of cell membranes. Due to the short duration of the spike the charge transfer is very small. The activation time (time of no return) of nerve cell membranes leading to an action potential is measured and found to be unexpectedly short. It can become as short as 3 microseconds for a voltage step of 10 V or higher.
💡 Research Summary
The paper introduces a novel extracellular stimulation paradigm that relies on the brief current spike generated when a voltage step is applied to living tissue. Unlike conventional stimulation methods that use prolonged current pulses or sustained electric fields, this approach exploits the rapid transient current that arises from the abrupt change in electric field (the Maxwell displacement current), followed by ionic charge redistribution and final membrane charging. The authors demonstrate the feasibility of this method by stimulating the human tongue, a tissue rich in sensory nerve endings, and measuring the charge‑injection thresholds for voltage steps ranging from 2 V to 20 V.
Key experimental observations include: (1) the current spike has a half‑width of roughly 1 µs, far shorter than typical stimulation pulses; (2) the spike’s decay is non‑exponential and can be decomposed into three distinct phases—(i) a Maxwell phase dominated by displacement current, (ii) a charge‑rearrangement phase where ionic currents flow through the conductive extracellular matrix, and (iii) a membrane‑charging phase in which the cell membrane acts as a capacitor completing the depolarization; (3) despite the ultra‑short duration, the total charge transferred is only on the order of tens of nanocoulombs, yet it is sufficient to elicit a perceptual response; (4) the “activation time” – the interval after which the membrane’s depolarization becomes irreversible and an action potential will inevitably fire – can be as short as 3 µs for voltage steps of 10 V or higher. This activation time is dramatically shorter (by four to five orders of magnitude) than the milliseconds‑scale times reported for conventional stimulation.
Theoretical analysis based on Maxwell’s equations and equivalent circuit modeling explains why each phase contributes to membrane voltage change. The initial Maxwell phase provides a rapid voltage jump that directly drives the membrane potential; the subsequent ionic phase amplifies this change by allowing charge to flow through the extracellular medium; finally, the capacitive charging of the membrane consolidates the voltage shift, ensuring that voltage‑gated sodium channels open. The authors argue that the brevity of the stimulus minimizes electrochemical side effects such as electrode corrosion, tissue heating, and Faradaic reactions, making the method potentially safer for chronic applications.
Limitations are acknowledged. The shape and amplitude of the current spike depend strongly on the tissue’s electrical heterogeneity (conductivity, permittivity) and on the exact geometry of the electrode–tissue interface. Consequently, the same voltage step may produce different spike profiles in muscle, brain, or peripheral nerve tissue. Moreover, generating fast, high‑voltage steps requires specialized high‑speed switching circuitry, raising practical concerns about device complexity, power consumption, and safety certification for human use.
In conclusion, the study provides compelling evidence that ultra‑short, high‑amplitude voltage steps can generate current spikes capable of reliably stimulating excitable cells with minimal charge delivery. This finding opens new avenues for designing high‑efficiency neural prostheses, sensory feedback systems, and selective nerve block technologies. Future work should extend the methodology to other excitable tissues, systematically map the relationship between voltage‑step parameters and fiber‑type selectivity, and develop compact, clinically‑compatible hardware that can deliver precisely controlled voltage steps while ensuring patient safety.
Comments & Academic Discussion
Loading comments...
Leave a Comment