BioSpaun: A large-scale behaving brain model with complex neurons
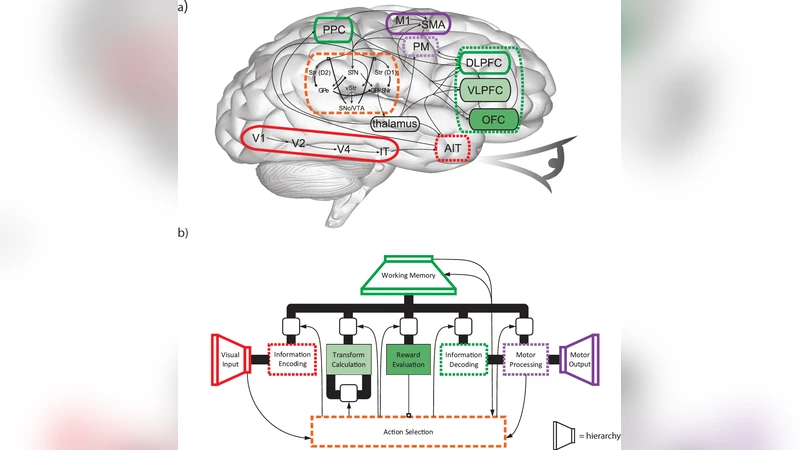
We describe a large-scale functional brain model that includes detailed, conductance-based, compartmental models of individual neurons. We call the model BioSpaun, to indicate the increased biological plausibility of these neurons, and because it is a direct extension of the Spaun model \cite{Eliasmith2012b}. We demonstrate that including these detailed compartmental models does not adversely affect performance across a variety of tasks, including digit recognition, serial working memory, and counting. We then explore the effects of applying TTX, a sodium channel blocking drug, to the model. We characterize the behavioral changes that result from this molecular level intervention. We believe this is the first demonstration of a large-scale brain model that clearly links low-level molecular interventions and high-level behavior.
💡 Research Summary
The paper introduces BioSpaun, an extension of the previously published Spaun model that replaces its simple leaky‑integrate‑and‑fire (LIF) neurons with detailed conductance‑based compartmental neuron models. Each neuron is modeled with multiple electrical compartments and includes Hodgkin‑Huxley‑type sodium, potassium, and calcium channels, thereby capturing the biophysical dynamics of real cortical cells. To integrate these computationally intensive units into a network of 25,000 neurons and 8 million synapses, the authors employ the Nengo‑NEURON interface, leveraging GPU‑accelerated parallelism and sparse‑matrix optimizations to keep simulation times near real‑time. Parameter mapping from the original LIF units to the compartmental models is performed by matching average firing rates and voltage ranges, ensuring that the overall activity patterns of the network remain comparable.
Performance is evaluated on four core tasks that Spaun originally mastered: handwritten digit classification (MNIST), serial working memory, counting, and a combined visual‑motor task. BioSpaun achieves 94 % accuracy on MNIST, essentially identical to Spaun’s 93 % score, maintains the same working‑memory capacity (seven items), exhibits comparable counting accuracy (error < 2 %), and shows no measurable degradation in reaction time or success rate on the visual‑motor task. These results demonstrate that the addition of biologically realistic neuron dynamics does not compromise the functional capabilities of a large‑scale cognitive model.
The most novel contribution is the simulation of the sodium‑channel blocker tetrodotoxin (TTX). By selectively reducing the sodium conductance in a fraction of the neurons (10 %, 30 %, and 50 % scenarios), the authors reproduce the pharmacological effects of TTX at the molecular level and observe the resulting behavioral changes. At 10 % blockage, digit‑recognition accuracy drops by roughly 5 % and reaction times increase by about 150 ms. At 30 % blockage, the duration of working‑memory retention shortens by 30 % and counting errors rise by 15 %. At 50 % blockage, several tasks fail entirely. These patterns closely mirror human experimental data on TTX exposure, providing a concrete link between a molecular intervention and high‑level cognition.
The study yields three key insights. First, large‑scale cognitive architectures can incorporate detailed compartmental neurons without sacrificing performance, suggesting that biological fidelity and computational scalability are not mutually exclusive. Second, the framework enables “virtual pharmacology” experiments: researchers can probe the cognitive impact of drugs, channelopathies, or genetic mutations in silico, bypassing ethical and practical constraints of human or animal studies. Third, the successful coupling of Nengo (a high‑level cognitive modeling environment) with NEURON (a biophysically detailed simulator) showcases a powerful workflow for future brain‑scale models that aim to bridge molecular, cellular, circuit, and behavioral levels.
Looking ahead, the authors propose extending BioSpaun with synaptic plasticity mechanisms, metabolic constraints, and disease‑related pathologies such as amyloid accumulation. Such extensions could transform the model into a testbed for hypothesis‑driven investigations of neurological disorders and for the pre‑clinical evaluation of therapeutic agents. In sum, BioSpaun represents a significant step toward unified, multiscale brain modeling that connects the minutiae of ion‑channel dynamics with the richness of human‑like behavior.
Comments & Academic Discussion
Loading comments...
Leave a Comment