Cancer and electromagnetic radiation therapy: Quo Vadis?
In oncology, treating cancer with a beam of photons is a well established therapeutic technique, developed over 100 years, and today over 50% of cancer patients will undergo traditional X-ray radiotherapy. However, ionizing radiation therapy is not the only option, as the high-energy photons delivering their cell-killing radiation energy into cancerous tumor can lead to significant damage to healthy tissues surrounding the tumor, located throughout the beam’s path. Therefore, in nowadays, advances in ionizing radiation therapy are competitive to non-ionizing ones, as for example the laser light based therapy, resulting in a synergism that has revolutionized medicine. The use of non-invasive or minimally invasive (e.g. through flexible endoscopes) therapeutic procedures in the management of patients represents a very interesting treatment option. Moreover, as the major breakthrough in cancer management is the individualized patient treatment, new biophotonic techniques, e.g. photo-activated drug carriers, help the improvement of treatment efficacy and/or normal tissue toxicity. Additionally, recent studies support that laser technology progresses could revolutionize cancer proton therapy, by reducing the cost of the needed installations. The aim of this review is to present some laser-based future objectives for cancer radiation therapy, aiming to address the relevant advances in the ionizing and non-ionizing radiation therapy, i.e. protons and heavy ions therapy, as well as photodynamic targeted and molecular therapies.
💡 Research Summary
The review examines the current state and future prospects of cancer radiation therapy, contrasting conventional ionizing X‑ray treatment with emerging non‑ionizing laser‑based approaches and particle‑based modalities such as proton and heavy‑ion therapy. Traditional radiotherapy, while effective at killing tumor cells, inevitably deposits dose along the beam path, causing collateral damage to surrounding healthy tissue. This inherent limitation has driven research into alternatives that can either spare normal tissue or enhance tumor specificity.
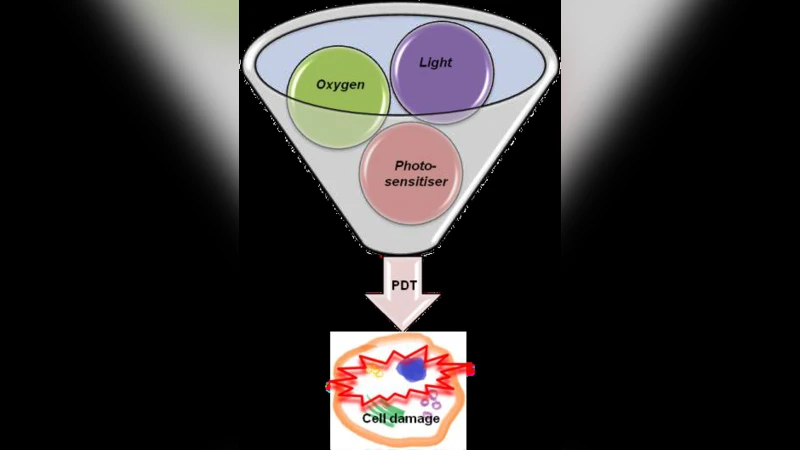
One major non‑ionizing strategy is photodynamic therapy (PDT). In PDT, a photosensitizer accumulated in the tumor is activated by a laser of a specific wavelength, generating reactive oxygen species that induce localized cell death. Because the cytotoxic effect is confined to the illuminated region, normal structures outside the light field receive minimal exposure. However, PDT efficacy depends on tumor oxygenation; hypoxic regions can blunt ROS production. Recent advances address this by designing oxygen‑independent photosensitizers and coupling them to nanocarriers that improve tumor uptake, penetration, and controlled release. These innovations broaden the applicability of PDT to otherwise resistant tumor microenvironments.
Particle therapy exploits the Bragg‑peak phenomenon, delivering the bulk of the particle’s kinetic energy at a precise depth that matches the tumor location. This results in steep dose fall‑off beyond the target, dramatically reducing normal‑tissue exposure. Despite its dosimetric superiority, clinical adoption is hampered by the massive size and cost of conventional cyclotrons or synchrotrons required to accelerate protons and heavy ions. Laser‑driven plasma accelerators (LPA) have emerged as a disruptive technology that can generate high‑energy ion beams using compact, high‑intensity laser pulses. Experimental demonstrations of 10 MeV protons producing clear Bragg peaks through tissue‑equivalent phantoms suggest that scaling to therapeutic energies (≈200 MeV for deep‑seated tumors) is within reach. If realized, LPA could shrink facility footprints, lower capital expenditures, and democratize access to particle therapy.
The authors also discuss synergistic regimens that combine laser‑mediated tumor modulation with particle irradiation. Laser‑induced vascular permeabilization, membrane depolarization, or transient disruption of DNA repair pathways can sensitize cancer cells to subsequent ionizing radiation, allowing dose de‑escalation while preserving—or even enhancing—tumor control probability. Such multimodal protocols align with the broader trend toward precision, personalized oncology, where treatment is tailored to the molecular and physiological characteristics of each patient’s disease.
Translational challenges remain. Optimizing laser parameters (pulse duration, repetition rate, wavelength) and particle beam characteristics (energy spread, spot size) is essential for reproducible, safe clinical outcomes. Pre‑clinical safety data, robust animal models, and early‑phase human trials are required to validate efficacy and establish toxicity thresholds. Regulatory pathways must evolve to accommodate hybrid devices that integrate photonic and particle components, as well as novel nanomedicines used for photo‑activation.
In summary, the review posits that non‑ionizing laser technologies—through refined photodynamic agents and nanocarrier systems—offer a route to highly selective tumor ablation with minimal normal‑tissue injury. Concurrently, laser‑driven plasma acceleration promises to overcome the economic and logistical barriers of conventional proton and heavy‑ion therapy. The convergence of these approaches could usher in a new era of cancer radiation treatment that is both more effective and more widely accessible, fulfilling the promise of individualized, minimally invasive oncologic care.
Comments & Academic Discussion
Loading comments...
Leave a Comment