Quantitative description of ion transport via plasma membrane of yeast and small cells
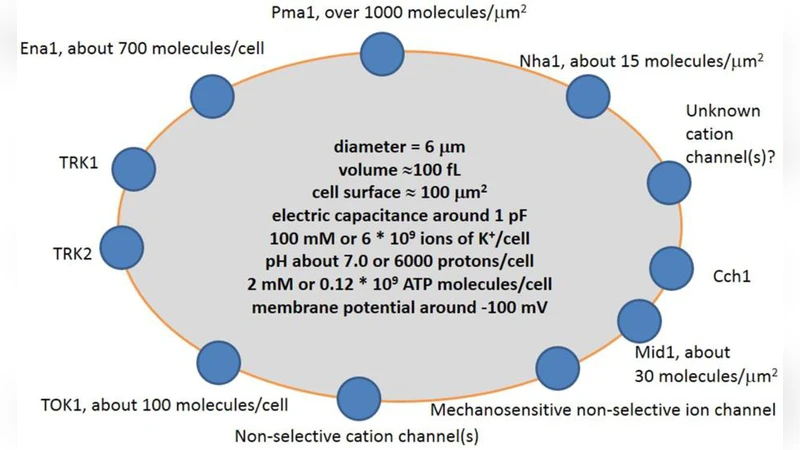
Modeling of ion transport via plasma membrane needs identification and quantitative understanding of the involved processes. Brief characterization of main ion transport systems of a yeast cell (Pma1, Ena1, TOK1, Nha1, Trk1, Trk2, non-selective cation conductance) and determining the exact number of molecules of each transporter per a typical cell allow us to predict the corresponding ion flows. In this review a comparison of ion transport in small yeast cell and several animal cell types is provided. The importance of cell volume to surface ratio is emphasized. The role of cell wall and lipid rafts is discussed in respect to required increase in spatial and temporal resolution of measurements. Conclusions are formulated to describe specific features of ion transport in a yeast cell. Potential directions of future research are outlined based on the assumptions.
💡 Research Summary
The paper provides a quantitative framework for understanding ion transport across the plasma membrane of Saccharomyces cerevisiae and compares it with several small animal cell types. By enumerating the exact copy numbers of the principal transport proteins—Pma1 (≈1 × 10⁶ per cell), Ena1 (1 × 10⁴‑10⁵), the voltage‑gated K⁺ channel TOK1 (≈5 × 10³), the Na⁺/H⁺ antiporter Nha1 (1 × 10³‑10⁴), the high‑affinity K⁺ uptake systems Trk1 and Trk2 (each 2‑5 × 10³), and a non‑selective cation conductance (estimated to contribute ~10 % of total membrane current)—the authors calculate the corresponding ion fluxes under physiological conditions. Using typical yeast dimensions (diameter ~5 µm, volume ~30 fL, surface area ~100 µm²), they derive a volume‑to‑surface ratio (V/S) of ~0.3 fL µm⁻², comparable to that of small animal cells such as erythrocytes, yet distinct because the yeast cell wall adds substantial electrical resistance and mechanical buffering.
The analysis shows that the dominant H⁺‑pumping activity of Pma1 generates a steady current of roughly 200 pA, establishing a membrane potential of –200 to –300 mV that drives secondary transport. Ena1 contributes about 20 pA of Na⁺ efflux under high‑salt stress, while TOK1 and the Trk systems together handle tens of picoamperes of K⁺ flux, sufficient to balance the charge moved by the proton pump. The non‑selective cation pathway, though poorly characterized, is inferred to provide a background leak that moderates extreme voltage excursions.
A central theme is the impact of the V/S ratio on ion homeostasis. In small cells, a given current produces a larger change in membrane potential than in larger cells; however, the yeast cell wall, composed of β‑glucans and mannoproteins, acts as a dielectric layer that dampens rapid voltage swings and buffers ion concentrations. Moreover, the authors highlight the role of lipid rafts—microdomains enriched in sphingolipids and sterols—where Pma1 and certain channels are preferentially localized. This spatial clustering raises the local protein density two‑ to three‑fold, creating micro‑electric fields that can accelerate ion exchange locally while preserving overall cellular stability.
Methodologically, the paper critiques current electrophysiological tools. Patch‑clamp recordings on yeast are limited by the tiny membrane area, leading to noise levels that obscure currents below ~10 pA. Fluorescent pH or voltage reporters lack the temporal resolution (<1 ms) needed to capture rapid transporter cycles. The authors therefore advocate for next‑generation approaches: nanofabricated electrodes capable of sub‑picoampere sensitivity, optogenetic actuators to trigger specific transporters with millisecond precision, and correlative cryo‑electron microscopy to map transporter distribution within rafts.
In the discussion, the authors compare yeast ion transport to that of small animal cells. Animal cells lack a rigid wall, so their membrane potential responds more directly to ion fluxes; however, they also possess sophisticated buffering systems (e.g., Na⁺/K⁺‑ATPase) that differ in kinetic parameters from yeast Pma1. The comparison underscores that while the fundamental physics of ion movement is universal, the cellular architecture (wall versus no wall, raft organization) dictates distinct regulatory strategies.
The conclusions summarize three specific features of yeast ion transport: (1) an exceptionally high density of the H⁺‑pump Pma1 that sets the electrochemical landscape; (2) the cell wall’s dual role as a mechanical protector and electrical damper; and (3) the functional compartmentalization of transporters within lipid rafts. Future research directions are outlined: detailed structural studies of raft‑associated complexes, development of single‑molecule current recordings, computational modeling of wall‑mediated ion diffusion, and systematic profiling of transporter expression under varied environmental stresses (high Na⁺, low pH, osmotic shock). By establishing a quantitative baseline, the work aims to bridge yeast physiology with broader cell‑biology questions and to inform biotechnological applications where precise ion control is essential.
Comments & Academic Discussion
Loading comments...
Leave a Comment