Hydrogen influence on generalized stacking fault of zirconium basal plane: a first-principles calculation study
The infuences of hydrogen on the generalized stacking fault (GSF) energies of the basal plane along the <10-10> and <11-20> directions in the hcp Zr were investigated using the first-principles calculation method. The modifications of the GSF energies were studied with respect to the different distances of H atoms away from the slip plane and hydrogen content there. The calculation results have shown that the GSF energies along the < 10-10 > direction drastically reduce when H atoms locate nearby the slip plane. But H atoms slightly decrease the GSF barrier for the <11-20> slipping case. Meanwhile, with the increase of hydrogen density around the slip plane, the GSF energies along both the two shift directions further reduced greatly. The physical origin of the reduction of GSF energies due to the existence of hydrogen atoms in Zr was analyzed based on the Bader charge method. It is interpreted by the Coulomb repulsion of the Zr atoms beside the slip plane due to the charge transfer from Zr to H .
💡 Research Summary
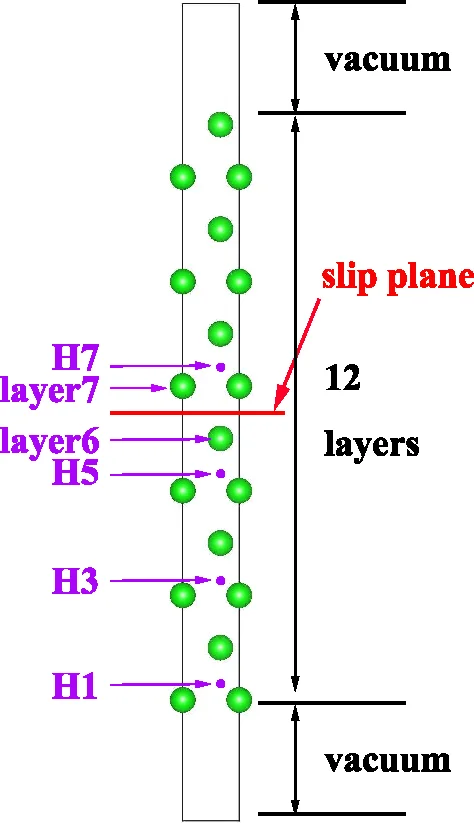
This paper investigates how hydrogen atoms affect the generalized stacking fault (GSF) energies of the basal {0001} plane in hexagonal close‑packed (hcp) zirconium using first‑principles density functional theory (DFT) calculations. Two slip directions, 1/3 <10‑10> and 1/3 <11‑20>, were examined. A 3 × 3 × 2 supercell containing 36 Zr atoms was constructed, and hydrogen was placed in the tetrahedral interstitial site (the most stable T‑site). Three configurations with a single H atom at different distances from the slip plane (H1, H3, H5) and a configuration with two H atoms (H57) were studied to assess both distance and concentration effects.
The pure Zr GSF energy maximum (γ_US) for the <10‑10> direction is 0.276 J m⁻², occurring at a fractional displacement of about 0.7–0.8. When H is far from the slip plane (H1, H3), γ_US decreases only slightly (≈5 %). However, when H is directly adjacent to the slip plane (H5), γ_US drops dramatically to 0.152 J m⁻² (≈45 % reduction) and the stable fault energy (γ_SF) falls to 0.039 J m⁻² (≈83 % reduction). Adding a second H atom (H57) further reduces γ_US to 0.070 J m⁻² and even makes γ_SF negative (‑0.115 J m⁻²), indicating that the fault becomes energetically favorable.
For the <11‑20> direction, the reductions are much smaller. The single‑H H5 configuration lowers γ_US from 0.449 J m⁻² to 0.407 J m⁻² (≈9 % reduction), and the H57 configuration yields γ_US = 0.285 J m⁻². The authors attribute this anisotropy to the larger inter‑layer spacing along <11‑20> (≈0.15 Å greater), which weakens the electrostatic interaction responsible for the energy drop.
Bader charge analysis reveals that each H atom gains about 0.79 e, while neighboring Zr atoms lose electrons (≈0.2 e per Zr for a single H, up to ≈0.6 e for the H57 case). The loss of electrons makes the Zr atoms on either side of the slip plane carry the same (negative) charge, leading to Coulomb repulsion that facilitates the relative displacement of the planes and thus lowers the GSF barrier. To validate this mechanism, the authors substituted H with C and N, which induce larger charge transfers and consequently produce even larger reductions in GSF energies, confirming the correlation between charge redistribution and fault energy.
The study concludes that hydrogen’s proximity to the basal slip plane and its concentration are key factors that reduce GSF energies in Zr, primarily through charge transfer‑induced Coulomb repulsion between Zr atoms. The effect is direction‑dependent, being more pronounced for <10‑10> than for <11‑20> due to geometric differences. These findings provide atomistic insight into how hydrogen embrittlement may evolve in zirconium cladding materials, suggesting that regions with higher hydrogen content will experience easier dislocation motion, potentially influencing plasticity and fracture behavior in light‑water reactor environments.
Comments & Academic Discussion
Loading comments...
Leave a Comment