Mechanism of dynamic reorientation of cortical microtubules due to mechanical stress
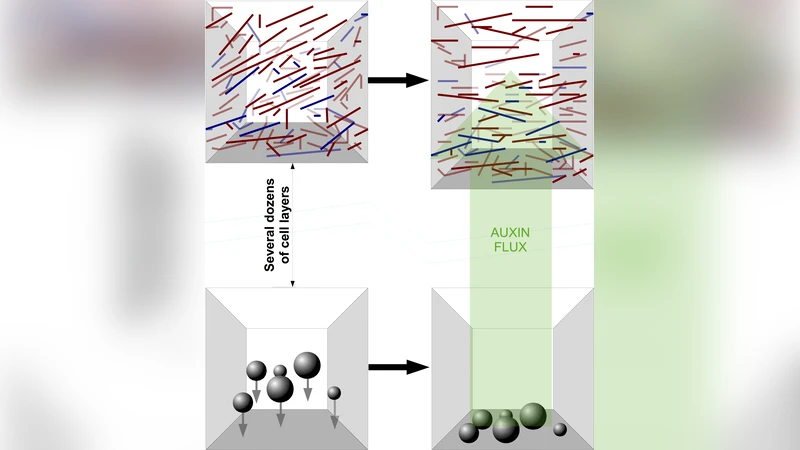
Directional growth caused by gravitropism and corresponding bending of plant cells has been explored since 19th century, however, many aspects of mechanisms underlying the perception of gravity at the molecular level are still not well known. Perception of gravity in root and shoot gravitropisms is usually attributed to gravisensitive cells, called statocytes, which exploit sedimentation of macroscopic and heavy organelles, amyloplasts, to sense the direction of gravity. Gravity stimulus is then transduced into distal elongation zone, which is several mm far from statocytes, where it causes stretching. It is suggested that gravity stimulus is conveyed by gradients in auxin flux. We propose a theoretical model that may explain how concentration gradients and/or stretching may indirectly affect the global orientation of cortical microtubules, attached to the cell membrane and induce their dynamic reorientation perpendicular to the gradients. In turn, oriented microtubules arrays direct the growth and orientation of cellulose microfibrils, forming part of the cell external skeleton and determine the shape of the cell. Reorientation of microtubules is also observed in reaction to light in phototropism and mechanical bending, thus suggesting universality of the proposed mechanism.
💡 Research Summary
The paper addresses a long‑standing gap in our understanding of how gravity perception in plants is translated into the mechanical re‑orientation of the cell wall that ultimately determines growth direction. Classical gravitropism models locate the perception in statocytes, where sedimenting amyloplasts generate an auxin redistribution that is thought to drive differential elongation in the distal growth zone. However, the link between this hormone gradient and the orientation of cortical microtubules (CMTs)—the scaffolds that guide cellulose microfibril deposition—has remained speculative.
The authors propose a quantitative, physics‑based model that couples auxin concentration gradients and membrane‑derived mechanical strain to the dynamic re‑orientation of CMTs. The model consists of three interconnected modules: (1) auxin transport, modeled by a one‑dimensional reaction‑diffusion equation that yields a steady‑state linear gradient ∇C as a result of amyloplast‑induced PIN polarity; (2) microtubule polymerisation/depolymerisation kinetics that are modulated by the magnitude of ∇C (polymerisation rate v = v0 exp(−α|∇C|), depolymerisation rate k = k0 + β|∇C|); and (3) a mechanical feedback where gravity‑induced cell‑wall tension generates a stress tensor σij that exerts a torque τ = γ σij n̂ × t̂ on CMTs through cross‑linking proteins such as MAP65.
By representing the local CMT orientation with a continuous angle field θ(x,t), the authors derive a phase‑field equation: ∂θ/∂t = D∇²θ – λ sin(2θ–2φ). Here φ denotes the direction of the auxin gradient or principal stress, λ quantifies the combined sensitivity to chemical and mechanical cues, and D is an effective angular diffusion constant. The equation possesses stable fixed points at θ = φ ± π/2, meaning that CMTs naturally align perpendicular to the gradient or stress direction. Numerical simulations show rapid convergence to this perpendicular alignment when λ/D exceeds a modest threshold (≈5), reproducing the experimentally observed swift re‑orientation of CMT arrays after gravistimulation.
The model’s predictions were benchmarked against published pharmacological data. Inhibition of auxin transport with NPA suppresses CMT re‑orientation, while microtubule destabilisation with oryzalin abolishes any alignment regardless of the presence of a gradient, consistent with the model’s dependence on both chemical and mechanical inputs. Moreover, the same framework accounts for phototropic CMT re‑orientation, where light‑induced auxin gradients replace gravity‑derived gradients, suggesting a universal mechanism for stimulus‑induced cell‑wall remodeling.
The authors discuss several limitations. The auxin field is treated as a static, linear gradient, ignoring possible feedback where CMT‑directed cellulose deposition alters wall stiffness and thus auxin flux. Only one class of cross‑linking protein (MAP65) is explicitly considered; other MAPs and motor proteins are subsumed into an effective torque coefficient γ. Despite these simplifications, the model provides clear, testable hypotheses: over‑expression of γ‑related cross‑linkers should accelerate re‑orientation, while reduction of λ (e.g., by mutating auxin‑responsive polymerisation regulators) should blunt the response.
In conclusion, the paper delivers a coherent mechanistic bridge between gravity‑induced auxin redistribution, mechanical strain, and the orientation of cortical microtubules. By showing that CMTs align perpendicular to the combined chemical‑mechanical cue, the work explains how differential cellulose deposition is orchestrated during gravitropism, phototropism, and mechanical bending. Future work involving real‑time auxin imaging, high‑speed super‑resolution microscopy, and CRISPR‑mediated manipulation of model parameters will be essential to validate and refine the proposed theory across diverse plant species and tissue types.
Comments & Academic Discussion
Loading comments...
Leave a Comment