Dissipation in noisy chemical networks: The role of deficiency
We study the effect of intrinsic noise on the thermodynamic balance of complex chemical networks subtending cellular metabolism and gene regulation. A topological network property called deficiency, known to determine the possibility of complex behavior such as multistability and oscillations, is shown to also characterize the entropic balance. In particular, only when deficiency is zero does the average stochastic dissipation rate equal that of the corresponding deterministic model, where correlations are disregarded. In fact, dissipation can be reduced by the effect of noise, as occurs in a toy model of metabolism that we employ to illustrate our findings. This phenomenon highlights that there is a close interplay between deficiency and the activation of new dissipative pathways at low molecule numbers.
💡 Research Summary
The paper investigates how intrinsic stochasticity influences the thermodynamic balance of complex chemical reaction networks (CRNs) that underlie cellular metabolism and gene regulation. The authors focus on a topological invariant of a CRN known as deficiency (δ), defined as the difference between the dimensions of the null spaces of the stoichiometric matrix (∇) and the incidence matrix (∂) of the complex‑graph representation. Deficiency counts the number of “hidden” closed reaction pathways that cannot be expressed as cycles in the complex graph.
Using the Anderson‑Craciun‑Kurtz (ACK) theorem, the authors show that when δ = 0 the stationary distribution of the stochastic dynamics (governed by the Chemical Master Equation) is of product‑form Poisson. In this case the average reaction flux ⟨v_ρ⟩ coincides with the deterministic mass‑action flux v_ρ(x), and consequently the average stochastic entropy production rate (EPR) σ equals the deterministic EPR σ̂. The difference δσ = σ − σ̂, termed the correlation EPR, therefore vanishes for deficiency‑zero networks.
When δ > 0 the stationary distribution is generally non‑Poissonian, leading to non‑trivial correlations among species. The authors derive an exact expression for δσ in steady state (Eq. 20‑21), showing that it is a weighted sum of moments of the population distribution multiplied by the thermodynamic forces G_ρ = ln(k⁺_ρ/k⁻_ρ). Thus δσ quantifies how stochastic fluctuations modify the entropy flow relative to the deterministic prediction.
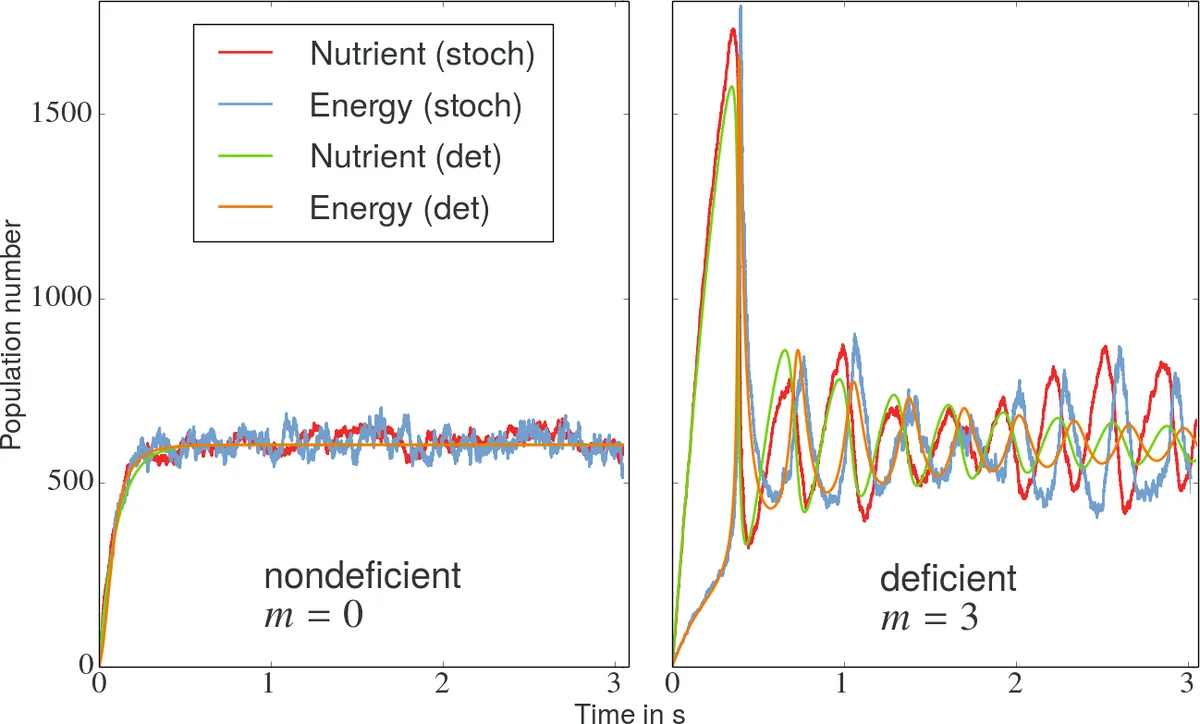
To illustrate these results, a minimal metabolic toy model is introduced: a three‑step cycle involving nutrients (N), energy tokens (E), and waste (W). The parameter m controls whether the second reaction includes an autocatalytic term; m = 0 yields δ = 0, while m > 0 gives δ = 1. Stochastic simulations (Gillespie algorithm) and deterministic ODE integration are performed for both cases. For δ = 0 the stochastic trajectories merely add structureless noise to the deterministic trajectory, and the measured EPRs coincide. For δ = 1, however, low molecule numbers suppress certain irreversible cycles, leading to a reduction of the stochastic EPR below its deterministic counterpart. This demonstrates that intrinsic noise can actually lower dissipation in deficient networks.
The paper also discusses linear CRNs (each reaction changes at most one molecule of each species). If the initial condition is Poissonian, the distribution remains Poissonian for all times, and δσ stays zero, extending earlier results by Mou et al. to the broader class of deficiency‑zero networks, whether linear or nonlinear.
In summary, the work establishes a direct link between a purely topological property of a reaction network (deficiency) and its nonequilibrium thermodynamic performance. Deficiency‑zero networks behave thermodynamically like their deterministic counterparts, while networks with positive deficiency can exhibit noise‑induced reductions in entropy production due to the activation or deactivation of hidden dissipative pathways at low copy numbers. This insight has implications for the design of efficient synthetic biochemical circuits and for understanding how natural metabolic pathways may exploit stochasticity to improve energetic efficiency.
Comments & Academic Discussion
Loading comments...
Leave a Comment