A role for ATP-dependent chromatin remodeling in the hierarchical cooperativity between noninteracting transcription factors
Chromatin remodeling machineries are abundant and diverse in eukaryotic cells. They have been involved in a variety of situations such as histone exchange and DNA repair, but their importance in gene expression remains unclear. Although the influence of nucleosome position on the regulation of gene expression is generally envisioned under the quasi-equilibrium perspective, it is proposed that given the ATP-dependence of chromatin remodeling enzymes, certain mechanisms necessitate non-equilibrium treatments. Examination of the celebrated chromatin remodeling system of the mouse mammary tumor virus, in which the binding of transcription factors opens the way to other ones, reveals that breaking equilibrium offers a subtle mode of transcription factor cooperativity, avoids molecular trapping phenomena and allows to reconcile previously conflicting experimental data. This mechanism provides a control lever of promoter responsiveness to transcription factor combinations, challenging the classical view of the unilateral influence of pioneer on secondary transcription factors.
💡 Research Summary
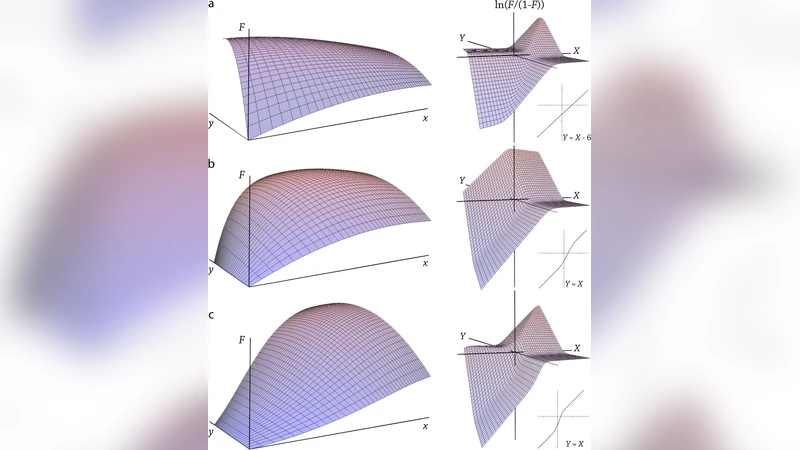
The paper investigates how ATP‑dependent chromatin remodeling contributes to cooperative interactions between transcription factors (TFs) that do not physically interact, using the mouse mammary tumor virus (MMTV) promoter as a model system. Historically, the activation of the MMTV promoter has been described as a hierarchical process: the glucocorticoid receptor (GR) binds first, remodels nucleosomes, and then NF1/Oct‑1 gains access to its sites, leading to synergistic transcriptional activation. However, subsequent studies reported conflicting observations—NF1/Oct‑1 can be pre‑bound before GR, and the promoter can remain inactive despite the presence of NF1/Oct‑1. These discrepancies suggest that a simple equilibrium‑based description is insufficient.
The authors first construct a conventional equilibrium model that assumes all binding steps are microscopically reversible. In this framework, the fractional promoter activity (F) is expressed as a weighted sum of the activities of GR (A) and NF1/Oct‑1 (B), with probabilities p(A) and p(B) derived from Adair equations. This model predicts “trapping” phenomena: once B binds, A cannot dissociate, or vice‑versa, which contradicts experimental evidence that GR can rapidly dissociate (FRAP half‑time ~12 ms) and that NF1/Oct‑1 may be present on DNA without activating transcription.
To resolve these contradictions, the authors propose a non‑equilibrium model that explicitly incorporates the ATP‑driven activity of SWI/SNF chromatin‑remodeling complexes. They define two chromatin states: an open state (P) and a closed state (P₀). GR can bind both states but with different equilibrium constants (K_A for P, K₀_A for P₀). NF1/Oct‑1 can bind only to the closed state (P₀). The transition from P₀ to P (opening) occurs at rate k_o, while the reverse transition (closing) occurs at rate k_c. Because these transitions consume ATP, they are treated as effectively irreversible on the timescale of TF binding.
Using rapid equilibration of TF‑DNA interactions and steady‑state balance for the chromatin transitions, the authors derive analytical expressions for the occupancies of each promoter state. The key results are:
- p(A) = (K_A
Comments & Academic Discussion
Loading comments...
Leave a Comment