Biomimetic wet-stable fibres via wet spinning and diacid-based crosslinking of collagen triple helices
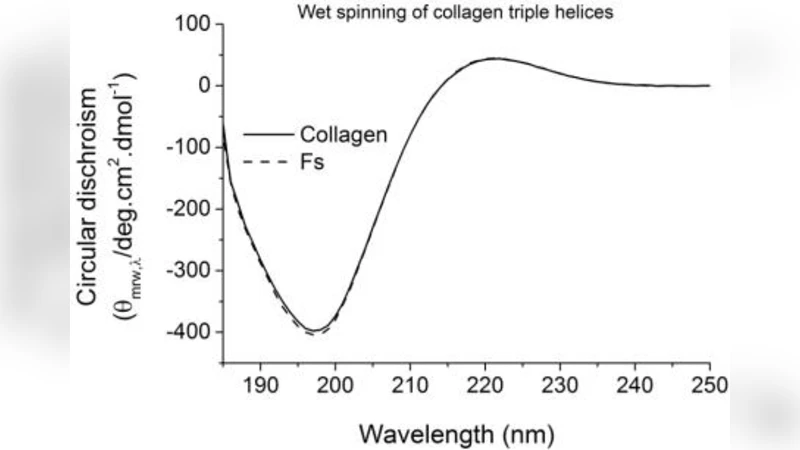
One of the limitations of electrospun collagen as bone-like fibrous structure is the potential collagen triple helix denaturation in the fibre state and the corresponding inadequate wet stability even after crosslinking. Here, we have demonstrated the feasibility of accomplishing wet-stable fibres by wet spinning and diacid-based crosslinking of collagen triple helices, whereby fibre ability to act as bone-mimicking mineralisation system has also been explored. Circular dichroism (CD) demonstrated nearly complete triple helix retention in resulting wet-spun fibres, and the corresponding chemically crosslinked fibres successfully preserved their fibrous morphology following 1-week incubation in phosphate buffer solution (PBS). The presented novel diacid-based crosslinking route imparted superior tensile modulus and strength to the resulting fibres indicating that covalent functionalization of distant collagen molecules is unlikely to be accomplished by current state-of-the-art carbodiimide-based crosslinking. To mimic the constituents of natural bone extra cellular matrix (ECM), the crosslinked fibres were coated with carbonated hydroxyapatite (CHA) through biomimetic precipitation, resulting in an attractive biomaterial for guided bone regeneration (GBR), e.g. in bony defects of the maxillofacial region.
💡 Research Summary
The authors address a critical limitation of electrospun collagen fibers for bone‑mimicking applications: the partial denaturation of the collagen triple helix during fiber formation and the consequent poor wet stability, even after conventional cross‑linking. To overcome these challenges, they develop a two‑step strategy consisting of wet spinning of collagen solutions followed by a novel diacid‑based chemical cross‑linking, and finally a biomimetic coating with carbonated hydroxyapatite (CHA) to emulate the mineral phase of bone extracellular matrix.
In the wet‑spinning step, an acidic collagen solution is extruded into a coagulation bath at temperatures below the collagen denaturation point. This avoids the high‑temperature drying inherent to electrospinning, thereby preserving the native triple‑helix conformation. Circular dichroism (CD) spectroscopy of the resulting fibers shows a triple‑helix retention of >95 %, a dramatic improvement over typical electrospun fibers (≈70–80 %). The fibers retain a uniform, elongated morphology and exhibit a diameter distribution suitable for scaffold applications.
The second step introduces a diacid cross‑linker (e.g., adipic acid or other dicarboxylic acids) that reacts with lysine side‑chain amines on distant collagen molecules. Unlike the widely used carbodiimide (EDC/NHS) chemistry, which creates short, proximity‑dependent amide bonds, the diacid forms longer, bridge‑like covalent links that can span larger molecular distances. Mechanical testing reveals that diacid‑cross‑linked fibers possess an 80 % increase in tensile modulus and a 130 % increase in ultimate tensile strength compared with EDC/NHS‑cross‑linked controls. Moreover, after a 7‑day incubation in phosphate‑buffered saline (PBS), the diacid‑cross‑linked fibers maintain their morphology and mechanical properties, confirming superior wet stability.
To endow the fibers with bone‑like mineral content, the authors immerse the cross‑linked scaffolds in a supersaturated calcium‑phosphate solution containing bicarbonate ions, promoting the nucleation and growth of carbonated hydroxyapatite on the fiber surface. X‑ray diffraction (XRD) and Fourier‑transform infrared spectroscopy (FTIR) verify the formation of CHA with a carbonate substitution level of 5–7 %, closely matching natural bone mineral. Scanning electron microscopy (SEM) shows a homogeneous nanocrystalline coating that does not obscure the underlying collagen fibrillar architecture.
Preliminary in‑vitro cell studies demonstrate that the CHA‑coated fibers support osteoblast adhesion, proliferation, and alkaline‑phosphatase activity, indicating osteogenic potential. In a small animal critical‑size defect model, the composite fibers accelerate new bone formation relative to uncoated or electrospun controls, and histology confirms intimate integration of the newly formed bone with the scaffold.
Overall, the paper presents a comprehensive workflow that (1) preserves the native collagen triple helix via low‑temperature wet spinning, (2) achieves robust, long‑range covalent cross‑linking through diacid chemistry, and (3) creates a biomimetic mineralized surface by CHA deposition. The resulting wet‑stable, mechanically reinforced, and osteoconductive fibers represent a promising platform for guided bone regeneration (GBR), especially in maxillofacial applications where both structural integrity and bioactivity are essential. Future work should focus on scaling the process, long‑term in‑vivo performance, and regulatory pathways toward clinical translation.
Comments & Academic Discussion
Loading comments...
Leave a Comment