Towards rational design of catalysts supported on a topological insulator substrate
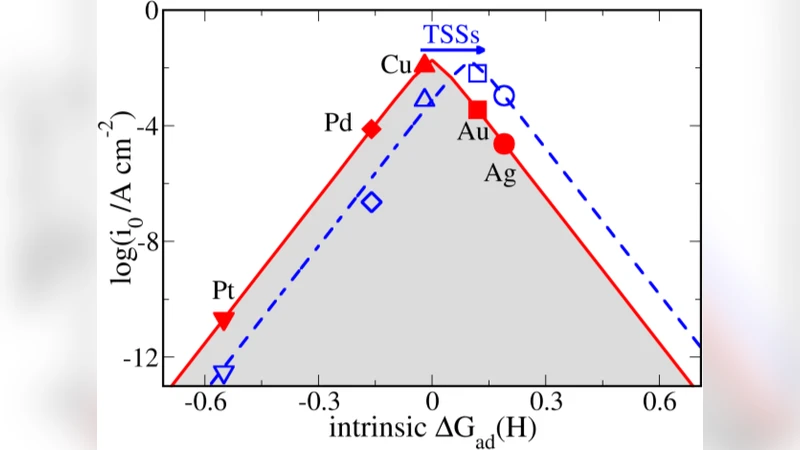
Exotic and robust metallic surface states of topological insulators (TIs) have been expected to provide a promising platform for novel surface chemistry and catalysis. However, it is still an unprecedented field how TIs affect the activity of catalysts. In this work, we study the effects of topological surface states (TSSs) on the activity of transition metal clusters (Au, Ag, Cu, Pt, and Pd), which are supported on a TI Bi2Se3 substrate. It was found the adsorption energy of oxygen on the supported catalysts can be always enhanced due to the TSSs. However, it does not necessarily mean an increase of the activity in catalytic oxidation reaction. Rather, the enhanced adsorption behavior in the presence of TSSs exhibits dual effects, determined by the intrinsic reactivity of these catalysts with oxygen. For the Au case, the activity of catalytic oxidation can be improved because the intrinsic binding between Au and O is relatively weak. In contrast, a negative effect is found for the Pt and Pd clusters since the intrinsic binding of Pt and Pd with oxygen is too strong. We also found that the effect of TSSs on the activity of hydrogen evolution reaction (HER) is quite similar, i.e. the metals with original weak reactivity can gain a positive effect from TSSs. The present work can pave a way for more rational design and selection of catalysts when using TIs as substrates.
💡 Research Summary
This paper investigates how the topological surface states (TSS) of a topological insulator (TI) substrate influence the catalytic performance of transition‑metal clusters. Using Bi₂Se₃(111) as the model TI, the authors performed density‑functional theory calculations on Au, Ag, Cu, Pt, and Pd nanoclusters supported on the surface. They compared the adsorption energies of atomic oxygen and hydrogen with and without the inclusion of TSS. The results show that TSS universally increase the binding strength of O and H on all metals, reflecting a TSS‑induced redistribution of electronic density that makes the metal surface more electron‑rich.
However, the impact on catalytic activity is not monotonic. For oxidation reactions, Au—whose intrinsic Au–O bond is weak—benefits from the stronger O adsorption, leading to a lower activation barrier and higher turnover frequency. In contrast, Pt and Pd already bind oxygen very strongly; the additional strengthening caused by TSS makes the oxygen‑containing intermediates too stable, hindering product desorption and thus decreasing overall activity. Ag and Cu, which sit in an intermediate regime, display modest or mixed effects. A similar dual behavior is observed for the hydrogen evolution reaction (HER). Metals with originally weak H* binding (Au, Ag) are pushed toward the optimal free‑energy window (ΔG_H* ≈ 0) by TSS, enhancing HER rates. Conversely, Pt and Pd, which already have strong H* adsorption, experience over‑binding that suppresses hydrogen release, reducing HER performance.
The authors conclude that TSS act as a “double‑edged sword”: they can either improve or impair catalytic function depending on the intrinsic metal–adsorbate interaction strength. Consequently, rational catalyst design on TI substrates must (1) select metals whose native binding energies are sub‑optimal so that TSS can bring them into the Sabatier‑optimal range, (2) employ accurate electronic‑structure models to predict the magnitude of TSS‑induced shifts, and (3) control the TI surface quality (layer thickness, defect density) and metal cluster size to fine‑tune the metal–TSS coupling. The study also suggests future work on other TI materials (e.g., Bi₂Te₃, Sb₂Te₃), the role of spin‑texture in reaction selectivity, and experimental validation through electrochemical and surface‑science techniques. Overall, the work provides a clear framework for exploiting topological surface states to engineer next‑generation catalysts with tailored activity and selectivity.