A model of chloroplast growth regulation in mesophyll cells
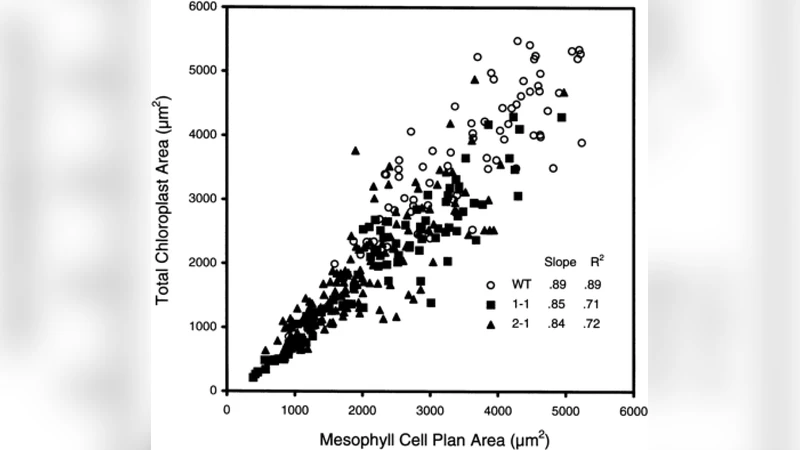
Chloroplasts regulate their growth to optimize photosynthesis. Quantitative data shows that the ratio of total chloroplast area to mesophyll cell area is constant across different cells within a single species, and also across species. Wild-type chloroplasts exhibit little scatter around this trend; highly irregularly-shaped mutant chloroplasts exhibit more scatter. Here we propose a model motivated by a bacterial quorum-sensing model consisting of a switch-like signalling network that turns off chloroplast growth. We calculated the dependence of the location of the relevant saddle-node bifurcation on the geometry of the chloroplasts. Our model exhibits a linear trend, with linearly growing scatter dependent on chloroplast shape, consistent with the data. When modelled chloroplasts are of a shape that grows with a constant area to volume ratio (disks, cylinders) we find a linear trend with minimal scatter. Chloroplasts with area and volume that do not grow proportionally (spheres) exhibit a linear trend with additional scatter.
💡 Research Summary
The paper addresses a striking regularity observed in leaf mesophyll cells: the total plan (projected) area occupied by chloroplasts scales linearly with the plan area of the host cell, and the proportionality constant is remarkably consistent across different cells of a species, across mutants, and even across plant species. Moreover, the scatter around this linear relationship grows roughly proportionally to cell size, and mutants with irregularly shaped chloroplasts display markedly larger scatter.
To explain these phenomena, the authors adapt a quorum‑sensing switch model originally developed for bacteria (Dockery & Keener, 2001). The core of the model consists of two ordinary differential equations describing the dynamics of an auto‑inducer molecule A and a regulatory protein R inside each chloroplast. Production of A is positively regulated by the R·A complex, creating a bistable switch: at low chloroplast density the system resides on a low‑A steady state, but as chloroplasts grow and become denser the effective degradation/secretion parameter d decreases, eventually reaching a saddle‑node bifurcation at d = d*. Crossing this bifurcation drives A above a threshold A*, which the authors interpret as the signal that shuts off chloroplast growth.
A key innovation is the explicit incorporation of cell and chloroplast geometry into the bifurcation parameter d. The mesophyll cell is modeled as two thin cytosolic layers (thickness τ) sandwiching a large vacuole; each layer has the same top surface area S, so the cell volume is V = 2Sτ and the plan area is S. Chloroplasts are idealized as one of three shapes—cylinders, thin disks, or spheres—each characterized by a single growth dimension ℓ (length, radius, or radius respectively) while the other dimensions are fixed by τ. For each shape the surface area S_c(ℓ) and volume V_c(ℓ) are known analytically, allowing the chloroplast volume fraction ρ = nV_c/(2Sτ) and the effective membrane permeability δ = nS_c·˜δ to be expressed directly in terms of ℓ, the number of chloroplasts n, and the cell size S.
Substituting these geometric expressions into the original quorum‑sensing equations yields a compact expression for d(ℓ,n,S). The condition d = d* defines a curve in the (ℓ,n) plane for each fixed S; solving this curve (subject to the physical constraint nV_c < 2Sτ) provides the set of chloroplast sizes and numbers at which growth stops. By converting each (ℓ,n) pair into a total chloroplast plan area (e.g., for disks A_total = n·πℓ²) and plotting A_total versus S, the model generates a “plan‑area plane” that can be directly compared with experimental data.
The analysis shows that when chloroplasts maintain a constant area‑to‑volume ratio—as is the case for cylinders and thin disks—the d* curve is essentially linear, leading to a tight linear relationship between total chloroplast area and cell area with minimal scatter. By contrast, spherical chloroplasts have surface area scaling as ℓ² while volume scales as ℓ³; this mismatch causes the d* curve to bend, allowing a broader range of (ℓ,n) combinations for a given S and thereby producing larger scatter. This geometric dependence reproduces the empirical observation that mutants with irregular, more three‑dimensional chloroplasts (arc2, arc3) exhibit greater variability.
Furthermore, the model predicts that the magnitude of scatter should increase roughly linearly with cell size because d* contains a term inversely proportional to S. Larger cells therefore experience a steeper slope in the (ℓ,n) space, amplifying the effect of discrete chloroplast numbers on the total plan area. This matches the observed proportional increase in variance with cell plan area.
Parameter sensitivity analysis indicates that the effective membrane permeability ˜δ and the extracellular degradation rate k_E strongly influence the location of d*. Higher permeability (or lower extracellular degradation) lowers d* more rapidly as chloroplasts accumulate, causing the growth‑stop switch to fire at lower chloroplast numbers. Conversely, increasing the intracellular degradation rate k_A shifts d* upward, allowing more chloroplasts to accumulate before the switch is triggered.
In summary, the authors provide a parsimonious yet biologically plausible mathematical framework that links chloroplast geometry, population density, and a bistable quorum‑sensing‑like switch to the experimentally observed linear scaling of chloroplast plan area with cell plan area and the shape‑dependent scatter around this trend. The model not only captures the essential features of wild‑type data but also explains why mutants with altered chloroplast morphology deviate more strongly. By reducing a complex intracellular signaling network to two ODEs while retaining explicit geometric dependence, the work offers a clear mechanistic hypothesis that can be tested experimentally—e.g., by manipulating chloroplast surface‑to‑volume ratios or membrane permeability—and sets the stage for future investigations into the molecular identity of the putative chloroplast quorum‑sensing pathway.
Comments & Academic Discussion
Loading comments...
Leave a Comment