Optical control of waves in a cardiac excitable medium
In nature, excitable reaction-diffusion systems found in diverse settings (e.g. chemical reactions, metal rust, yeast, amoeba, heart, brain) generate geometrically similar macroscopic waves(1,2). For the heart and brain, the spatiotemporal patterns formed by these excitation waves separate healthy from diseased states(1-3). Current electrical and pharmacological therapies for bioelectric disorders often lack the necessary spatiotemporal precision needed to control these patterns. Optical methodologies have the potential to overcome these limitations, but have only been demonstrated in simple systems, e.g. the Belousov-Zhabotinsky (BZ) chemical reaction4. Here we combine novel dye-free optical imaging with optogenetic actuation for dynamic control of cardiac excitation waves. We leverage patterned light to optically control emergent macroscopic properties of cardiac tissue: wave direction, wave speed, and spiral wave chirality. This all-optical approach offers a fundamentally new experimental platform for the study and control of pattern formation in complex biological excitable systems.
💡 Research Summary
The paper presents a fully optical platform that combines dye‑free high‑speed imaging with optogenetic stimulation to observe and manipulate excitation waves in cardiac tissue with unprecedented spatiotemporal precision. Traditional therapies for electrical disorders—such as implantable defibrillators, pacing electrodes, or pharmacological agents—act globally and lack the ability to target specific wave fronts or patterns. By eliminating fluorescent dyes, the authors avoid phototoxicity and signal interference, using reflected light captured by a high‑frame‑rate CMOS camera to infer transmembrane voltage changes across a two‑dimensional myocardial sheet. Simultaneously, they express the light‑gated ion channel CheR2 in cardiomyocytes and project patterned blue light (≈470 nm) via a digital micromirror device (DMD). The projected light locally depolarizes cells, allowing the researchers to dictate where a wave initiates, how it propagates, and even its chirality.
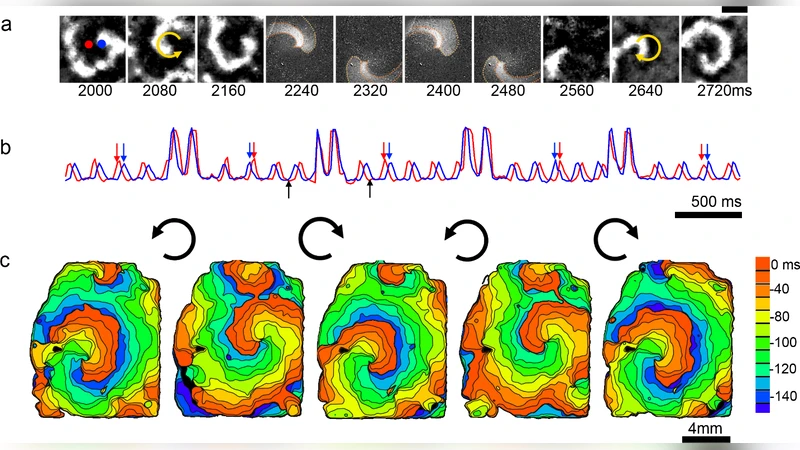
Key experimental demonstrations include: (1) “wave steering,” where a moving light stripe redirects a planar wave along a prescribed trajectory; (2) speed modulation, showing that increasing light intensity or expanding the illuminated region accelerates wave velocity up to a two‑fold increase, mirroring the effect of enhanced electrical conductivity in classical reaction‑diffusion models; and (3) chirality reversal of spiral waves, achieved by first suppressing the existing spiral core with a counter‑rotating light pattern and then re‑exciting the tissue with a pattern rotating in the opposite direction, thereby generating a new spiral with reversed rotation without any electrical shock or drug application.
These results demonstrate that the nonlinear diffusion‑reaction dynamics governing excitable media can be reshaped by dynamic optical boundary conditions. The approach is mathematically analogous to altering boundary terms in FitzHugh‑Nagumo or Aliev‑Panfilov equations, but it is realized experimentally in living tissue. Because imaging and stimulation share the same optical pathway, the system can be closed‑looped: real‑time image analysis can feed back to adjust light patterns on the fly, enabling adaptive control of wave fronts.
Beyond cardiac applications, the authors argue that any biological or chemical excitable system—neuronal cultures, cortical slices, slime‑mold aggregation, or Belousov‑Zhabotinsky reactions—could be subjected to similar all‑optical control, opening a new experimental arena for studying pattern formation, turbulence, and defect dynamics. Clinically, the technique suggests a minimally invasive alternative to conventional defibrillation: instead of delivering a high‑energy shock to the entire heart, a focused light pattern could extinguish or re‑orient pathological re‑entrant circuits (e.g., ventricular fibrillation) while sparing surrounding tissue.
Future work outlined includes scaling the method to three‑dimensional intact hearts, testing in large animal models, optimizing opsin expression and light delivery to reduce energy requirements, and integrating machine‑learning‑driven image processing for autonomous detection and correction of arrhythmic patterns. If successful, this all‑optical paradigm could transform both basic research on excitable media and the therapeutic management of bioelectric disorders.
Comments & Academic Discussion
Loading comments...
Leave a Comment