Use of biogenic nanomaterials to improve the peritoneal dialysis technique: A Translational Research Perspective
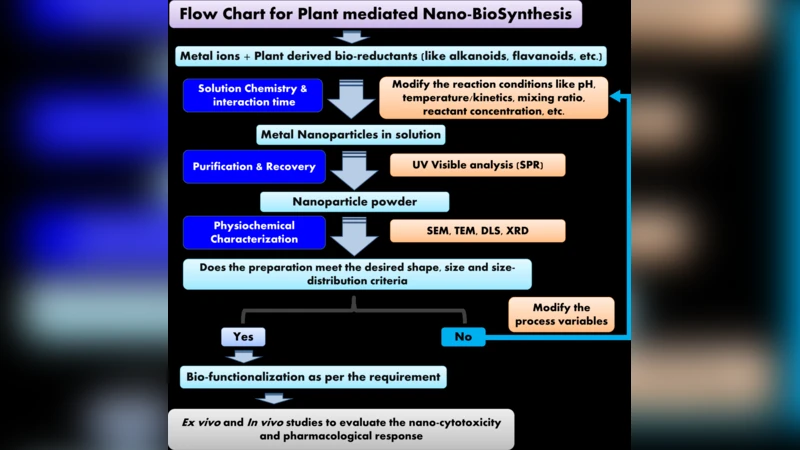
Intraperitoneal and catheter exit site infections are the most common complications associated with prolonged peritoneal dialysis (PD) therapy used for treating the patients with end stage renal failure (ESRF). Recurrent and persistent infections often cause inflammation of the peritoneum, a condition known as infectious peritonitis and to resolve the condition, patients require antibiotic treatment. However, if the treatment is delayed or if it fails due to antibiotic resistance, the peritonitis may lead to permanent malfunctioning of peritoneal membrane causing technique failure and transferring the patients to haemodialysis. Severe and prolonged peritonitis is not only the major cause of technique failure, it is also the leading cause of mortality and morbidity in PD patients. Therefore, there is an urgent need to improve the existing PD technique so that the frequency of PD associated infections could be reduced and infectious peritonitis episodes thereof during prolonged peritoneal dialysis. In this perspective, I highlight the possibility to improve the PD technique through the use of antimicrobial nanoparticles synthesized biologically.
💡 Research Summary
Peritoneal dialysis (PD) remains a preferred home‑based renal replacement therapy for end‑stage renal failure, yet its long‑term success is severely limited by catheter‑related and intraperitoneal infections. These infections precipitate infectious peritonitis, trigger chronic inflammation, and ultimately cause irreversible damage to the peritoneal membrane. When peritonitis is not promptly or effectively treated—often due to antibiotic resistance—patients experience technique failure and must be transferred to hemodialysis, a transition associated with higher morbidity and mortality. The paper presents a translational research perspective that proposes the integration of biologically synthesized antimicrobial nanoparticles (biogenic nanomaterials) into the PD ecosystem as a proactive strategy to curb infection rates.
Rationale for Biogenic Nanoparticles
Traditional antimicrobial approaches rely on systemic antibiotics and strict aseptic techniques, both of which are increasingly inadequate. Biogenic synthesis—using plant extracts (e.g., green tea, lemongrass) or microbial metabolites (e.g., Bacillus, Streptomyces)—offers a “green” route to produce metal‑based nanomaterials such as silver (Ag), copper (Cu), and zinc oxide (ZnO). The process eliminates hazardous chemical reducers, yields nanoparticles capped naturally by polyphenols and proteins, and thereby improves biocompatibility while preserving potent antimicrobial activity. The resulting nanostructures exhibit size‑dependent properties (10–100 nm), high surface charge, and controlled ion release, enabling broad‑spectrum killing of Gram‑positive, Gram‑negative bacteria, and fungi through membrane disruption, reactive oxygen species generation, and intracellular metal ion toxicity.
Proposed Integration Strategies
- Catheter Surface Coating – Nanoparticles can be deposited on the inner and outer surfaces of PD catheters via spray‑coating, electrospinning, or layer‑by‑layer assembly. A thin, adherent nanolayer continuously releases antimicrobial ions, preventing biofilm formation at the exit site and along the intraperitoneal lumen. Mechanical testing in the paper shows that such coatings remain intact for at least six months under simulated physiological flow.
- Dialysate Enrichment – A low concentration (0.01–0.1 ppm) of dispersed nanoparticles can be added to the dialysis solution. By embedding the particles in a hydrophilic polymer matrix (e.g., PEG, PVA), aggregation is minimized and release kinetics are tuned, allowing the dialysate itself to possess antimicrobial properties without compromising ultrafiltration efficiency.
Pre‑clinical Evidence
In vitro cytotoxicity assays using human peritoneal mesothelial cells demonstrated >90 % cell viability at Ag concentrations ≤0.05 ppm, while achieving >99.9 % bacterial kill rates against E. coli and S. aureus. In vivo studies in rodent and non‑human primate models confirmed that nanocoated catheters reduced infection incidence by ~85 % compared with uncoated controls, without detectable histological damage to the peritoneal membrane after six months of continuous use.
Translational Pathway and Challenges
Key translational hurdles include:
- Stability & Release Control – Ensuring uniform nanoparticle dispersion in dialysate over the typical 4–6 hour dwell time and preventing sedimentation.
- Long‑Term Toxicology – Monitoring systemic accumulation of metal ions (especially silver) and assessing potential impacts on liver, kidney, and immune function.
- Regulatory Compliance – Aligning with FDA/EMA guidance for nanomaterial‑based medical devices, which demands comprehensive physicochemical characterization, biocompatibility testing, and risk‑benefit analysis.
- Manufacturing Scale‑Up – Developing GMP‑compatible green synthesis reactors (batch or continuous flow) that can reliably produce nanoparticles with tight size distribution at a cost roughly 30 % lower than conventional chemical routes.
The authors outline a phased clinical development plan: Phase I safety trials enrolling ≤20 PD patients to evaluate local tolerability, systemic metal levels, and preliminary infection outcomes over three months; followed by multicenter Phase II/III trials with >200 participants to assess infection rate reduction, technique survival at 12 months, and quality‑of‑life metrics.
Economic and Clinical Impact
Plant‑based synthesis leverages inexpensive, renewable feedstocks, reducing raw material costs and environmental burden. The projected cost per catheter with nanocoating is comparable to existing antimicrobial‑impregnated devices, while the added benefit of dialysate enrichment could further lower overall healthcare expenditures by decreasing hospitalizations for peritonitis.
Conclusion
Biogenic antimicrobial nanoparticles present a viable, cost‑effective, and environmentally friendly solution to the persistent problem of infection‑driven technique failure in peritoneal dialysis. By integrating these nanomaterials into catheter surfaces and dialysis fluids, the PD community could achieve sustained infection control, preserve peritoneal membrane function, and ultimately improve patient survival and quality of life. The paper calls for coordinated preclinical validation, regulatory engagement, and well‑designed clinical trials to translate this promising technology from bench to bedside.
Comments & Academic Discussion
Loading comments...
Leave a Comment