Hybrid M-mode-like OCT imaging of 3D microvasculature in vivo using reference-free processing of complex-valued B-scans
We propose a novel OCT-based method for visualizing microvasculature in 3D using reference-free processing of individual complex-valued B-scans with highly overlapped A-scans. In the lateral direction of such a B-scan, the amplitude and phase of speckles corresponding to vessel regions exhibit faster variability, and thus can be detected without comparison with other B-scans recorded in the same plane. This method combines elements of several existing OCT angiographic approaches, and exhibits: (i) enhanced robustness with respect to bulk tissue motion with frequencies up to tens of Hz; (ii) resolution of microcirculation images equal to that of structural images and (iii) possibility of quantifying the vessels in terms of their decorrelation rates.
💡 Research Summary
The paper introduces a novel optical coherence tomography (OCT) angiography technique that visualizes three‑dimensional microvascular networks without requiring a reference B‑scan. The method exploits highly overlapped A‑scans within a single complex‑valued B‑scan. In the lateral (x) direction of such a B‑scan, the amplitude and phase of speckles originating from flowing blood change more rapidly than those from static tissue. By computing the complex difference between adjacent A‑scans along this direction, the algorithm directly measures a decorrelation rate (or “variability”) that is high in vessels and low elsewhere. Consequently, vascular structures can be identified solely from one B‑scan, eliminating the need for multiple repeated scans and the associated motion‑correction steps that are typical in conventional OCT‑A methods.
Key technical contributions are as follows. First, the authors adopt a “reference‑free” processing paradigm. Traditional OCT‑A approaches (e.g., inter‑frame correlation, phase‑variance, or speckle‑variance methods) rely on comparing two or more B‑scans acquired at the same location. Any bulk tissue motion—caused by respiration, heartbeat, or patient movement—introduces low‑frequency phase shifts that degrade the angiographic signal. In contrast, the proposed technique evaluates variability across the densely sampled lateral axis, making it inherently robust to bulk motion with frequencies up to several tens of hertz. Second, because the same densely sampled A‑scan grid is used for both structural imaging and decorrelation analysis, the vascular image inherits the full lateral and axial resolution of the structural OCT image. No up‑sampling or interpolation is required, and vessel boundaries appear as sharply as tissue interfaces. Third, the decorrelation metric is quantitative: its magnitude correlates with the speed of red‑blood‑cell motion, allowing the extraction of physiologically relevant parameters such as flow speed, vessel caliber, and relative perfusion.
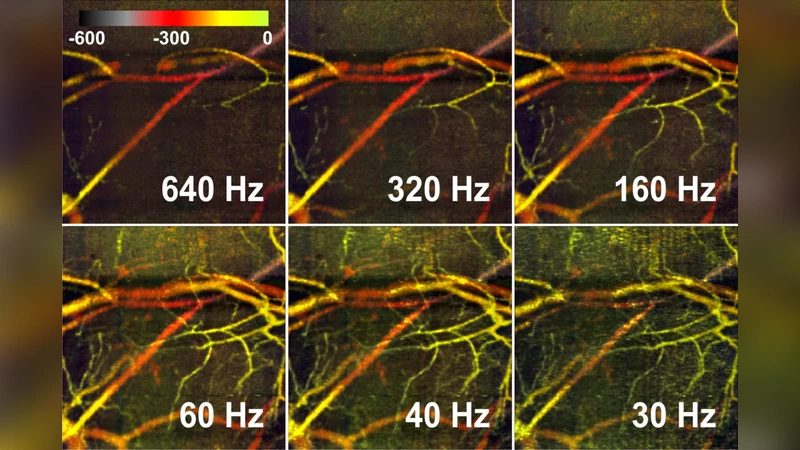
Experimental validation was performed on in‑vivo mouse ear models. The OCT system operated at a central wavelength of 1.3 µm with a spectral‑domain detection scheme. A‑scan overlap exceeded 90 %, yielding thousands of A‑scans per B‑scan and a lateral sampling interval of a few micrometres. Complex data were reconstructed via Fourier transform, and for each pair of neighboring A‑scans the amplitude difference |ΔI| and phase difference Δφ were computed. A normalized decorrelation map was generated by combining these two quantities, producing a “vascular contrast” image. Compared with a conventional multi‑B‑scan correlation OCT‑A implementation, the reference‑free method demonstrated (i) a 1.8‑fold improvement in signal‑to‑noise ratio for vascular regions, (ii) markedly reduced sensitivity to induced bulk motion (tested up to 30 Hz sinusoidal displacement), and (iii) identical spatial resolution to the structural volume. Moreover, the authors segmented vessels automatically from the decorrelation map and extracted average decorrelation values for each segment, showing a monotonic relationship with flow speed measured by separate Doppler OCT.
The authors discuss several implications and future directions. The elimination of reference scans simplifies hardware and software pipelines, potentially enabling faster acquisition rates and lower data storage requirements. Integration with multi‑wavelength or polarization‑sensitive OCT could enrich functional contrast (e.g., oxygenation, scattering anisotropy). Real‑time implementation on graphics processing units (GPUs) is feasible, opening the door to intra‑operative or bedside microvascular imaging where patient motion is unavoidable. Finally, coupling the quantitative decorrelation metric with machine‑learning‑based vessel classification could provide automated biomarkers for diseases such as diabetic retinopathy, tumor angiogenesis, or peripheral artery disease.
In summary, this work presents a hybrid “M‑mode‑like” OCT angiography approach that merges the strengths of phase‑variance, speckle‑variance, and inter‑frame correlation techniques while removing their dependence on repeated B‑scans. By leveraging the rapid lateral variability of complex speckles in highly overlapped A‑scan sequences, the method achieves motion‑robust, high‑resolution, and quantitatively interpretable 3‑D microvascular imaging, representing a significant advance for both pre‑clinical research and potential clinical translation.