Cs in high oxidation states and as a p-block element
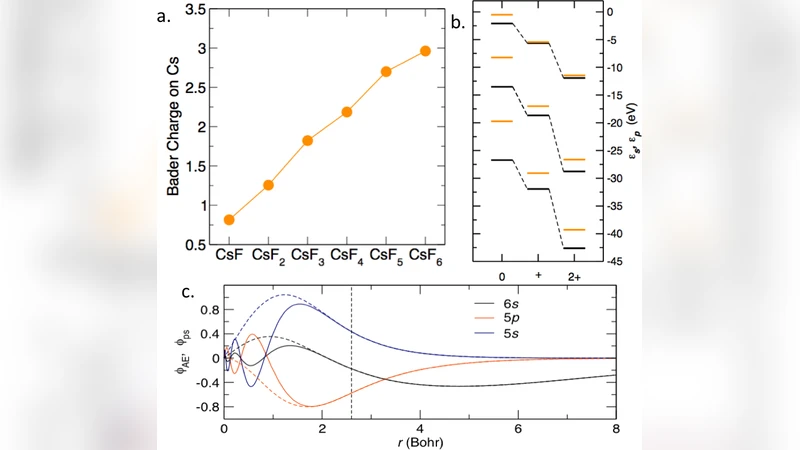
The major chemical feature of an element is the number of electrons available for forming chemical bonds. A doctrine rooted in the atomic shell model states that the atoms will maintain a complete inner shell while interacting with other atoms. Therefore, group IA elements, for example, are invariably stable in the +1 charge state because the p electrons of their inner shells do not react with other chemical species. This general rule governs our understanding of the structures and reactions of matter and has never been challenged. In this work, we show for the first time that while mixing with fluorine under pressure, Cs atoms will share their 5p electrons and become oxidized to a higher charge state. The formal oxidation state can be as high as +5 within the pressure range of our study (<200 GPa) and stable Cs2+ and Cs3+ compounds can form at lower pressures. While sharing its 5p electrons, Cs behaves like a p-block element forming compounds with molecular, covalent, ionic and metallic features. Considering the pressure range required for the CsFn compounds, the inner shell electrons in other group IA and IIA elements may also bond with F or other chemical species under higher pressure.
💡 Research Summary
The paper presents a groundbreaking investigation into the chemistry of cesium (Cs) under high‑pressure conditions, demonstrating that Cs can achieve oxidation states far beyond its canonical +1 state by engaging its inner‑shell 5p electrons in bonding with fluorine (F). Using a combination of diamond‑anvil cell experiments up to 200 GPa and state‑of‑the‑art first‑principles calculations (DFT with the USPEX structure‑search algorithm), the authors systematically explore the Cs–F binary system. Experimentally, new diffraction peaks appear once the pressure exceeds ~30 GPa, indicating the formation of previously unknown CsF₂, CsF₃, and CsF₅ phases. Raman spectroscopy corroborates structural changes and reveals mixed ionic‑covalent character. Computationally, the enthalpy landscape shows that CsF₂ and CsF₃ are thermodynamically favored at intermediate pressures (≈50–100 GPa), while CsF₅ becomes the most stable compound above ~100 GPa. Detailed electronic‑structure analyses—Bader charge partitioning, electron‑localization function (ELF), and projected density of states—demonstrate that the 5p orbitals of Cs are depopulated and participate directly in Cs–F bonds. In CsF₅, for instance, the Bader charge on Cs approaches +5, a formal oxidation state unprecedented for an alkali metal. The bonding motifs range from predominantly ionic (CsF₂) to highly covalent and even metallic (CsF₅), mirroring the diversity typical of p‑block elements. The authors argue that pressure compresses the electron cloud, reduces the energy gap between outer‑valence (6s) and inner‑valence (5p) shells, and effectively “activates” electrons that are inert at ambient conditions. This challenges the long‑standing atomic‑shell model that treats inner shells as chemically inert and confines group‑IA elements to a +1 oxidation state. The work also speculates that similar high‑oxidation‑state chemistry could be realized for other group‑IA and IIA elements (K, Rb, Ba, etc.) at even higher pressures, opening a new frontier in high‑pressure chemistry. Potential applications are discussed, including the design of novel superconductors, high‑energy‑density materials, and exotic catalysts that exploit the unique electronic configurations of high‑oxidation‑state alkali metals. In summary, the study provides compelling experimental and theoretical evidence that cesium can behave as a p‑block element under compression, fundamentally revising our understanding of oxidation states, electron participation, and the limits of chemical bonding in the periodic table.