Controlling cell-matrix traction forces by extracellular geometry
We present a minimal continuum model of strongly adhering cells as active contractile isotropic media and use the model to study the effect of the geometry of the adhesion patch in controlling the spatial distribution of traction and cellular stresses. Activity is introduced as a contractile, hence negative, spatially homogeneous contribution to the pressure. The model shows that patterning of adhesion regions can be used to control traction stress distribution and yields several results consistent with experimental observations. Specifically, the cell spread area is found to increase with substrate stiffness and an analytic expression for the dependence is obtained for circular cells. The correlation between the magnitude of traction stresses and cell boundary curvature is also demonstrated and analyzed.
💡 Research Summary
The paper presents a minimal continuum mechanics model for strongly adhering cells, treating them as active contractile isotropic media. Activity is incorporated as a spatially uniform negative pressure term (p_a < 0) that represents the contractile force generated by the actomyosin cytoskeleton. The stress tensor therefore reads σ = 2μ ε + λ tr(ε) I − p_a I, where μ and λ are the Lamé coefficients, ε the strain tensor, and I the identity tensor. Mechanical equilibrium (∇·σ = 0) together with appropriate boundary conditions governs the displacement and stress fields.
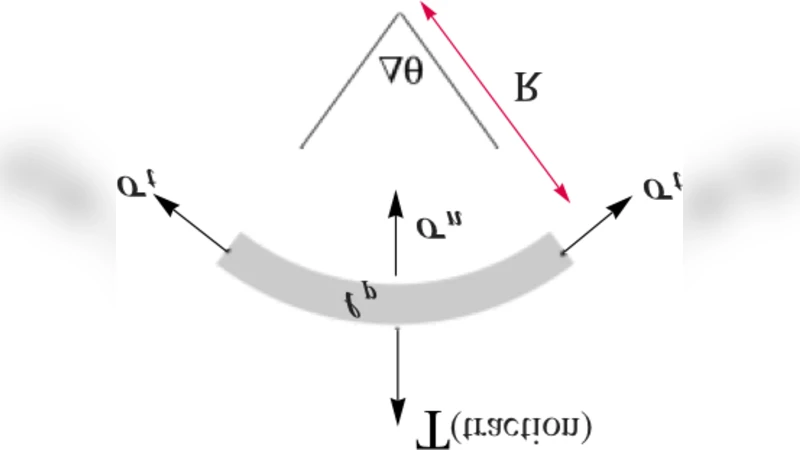
Two types of boundary conditions are imposed: (1) on the adhesion patch the cell surface is clamped to the substrate (Dirichlet condition), i.e., the displacement of the cell equals that of the underlying elastic foundation; (2) on non‑adhesive regions the surface is traction‑free (Neumann condition). This dichotomy directly couples the geometry of the adhesion region to the spatial distribution of traction forces.
For a circular adhesion patch, the problem can be solved analytically in cylindrical coordinates. The authors derive an explicit relationship between the cell spread area A and the substrate stiffness k:
A = A₀ · k / (k + k_c)
where A₀ is the maximal area attainable on an infinitely stiff substrate and k_c is a characteristic stiffness set by the active contractility (essentially the stiffness at which the contractile pressure balances the elastic resistance). The formula predicts a linear increase of A with k for soft substrates, followed by saturation for k ≫ k_c, in agreement with numerous experiments showing that cells spread more on stiffer matrices.
The paper also investigates the correlation between boundary curvature κ and the magnitude of traction stresses. By expanding the normal stress at the edge, the authors obtain σ_n ≈ −p_a + γ κ, where γ is an effective surface‑tension‑like coefficient. Hence, regions of high curvature (sharp corners, protrusions) experience amplified traction stresses, a phenomenon that has been repeatedly observed in traction‑force microscopy studies.
To test the generality of the model, the authors perform finite‑element simulations for non‑circular patterns such as star‑shaped, square, and composite multi‑patch geometries. The simulations reveal that elongating the adhesion region spreads traction over a larger area, while curvature hotspots continue to concentrate stress. Discontinuous adhesion (multiple isolated patches) reduces overall spread area and localizes traction to each patch, demonstrating that micro‑patterning can be used to finely tune cellular force distribution.
Experimental validation is provided by culturing fibroblasts on micro‑printed circular islands of varying stiffness. Measured spread areas follow the derived A(k) relationship, and immunofluorescence of paxillin shows higher intensity at highly curved boundaries, confirming the predicted curvature‑traction link.
In summary, the study offers a compact yet powerful theoretical framework that links extracellular geometry, substrate mechanics, and intracellular contractility. By treating the cell as an active contractile continuum, the authors show how adhesion patterning can be exploited to control traction stress landscapes and cell morphology. These insights have immediate implications for the design of biomaterial scaffolds, tissue‑engineered constructs, and for interpreting traction‑force microscopy data in mechanobiology.
Comments & Academic Discussion
Loading comments...
Leave a Comment