Spontaneous symmetry breaking in active droplets provides a generic route to motility
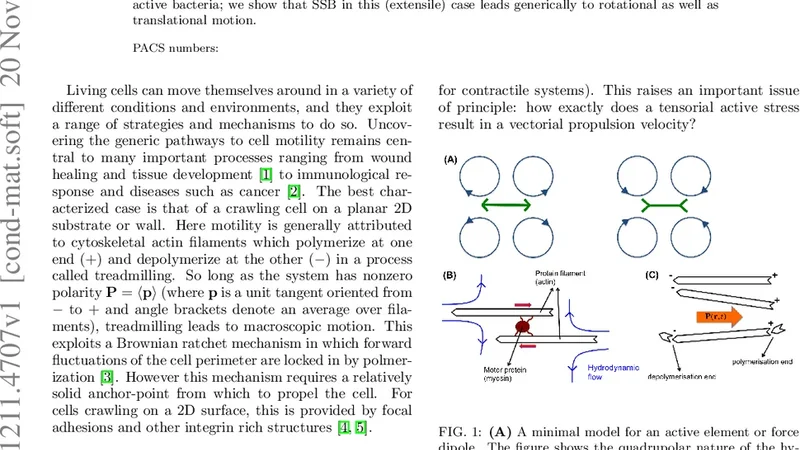
We explore a generic mechanism whereby a droplet of active matter acquires motility by the spontaneous breakdown of a discrete symmetry. The model we study offers a simple representation of a “cell extract” comprising, e.g., a droplet of actomyosin solution. (Such extracts are used experimentally to model the cytoskeleton.) Actomyosin is an active gel whose polarity describes the mean sense of alignment of actin fibres. In the absence of polymerization and depolymerization processes (’treadmilling’), the gel’s dynamics arises solely from the contractile motion of myosin motors; this should be unchanged when polarity is inverted. Our results suggest that motility can arise in the absence of treadmilling, by spontaneous symmetry breaking (SSB) of polarity inversion symmetry. Adapting our model to wall-bound cells in two dimensions, we find that as wall friction is reduced, treadmilling-induced motility falls but SSB-mediated motility rises. The latter might therefore be crucial in three dimensions where frictional forces are likely to be modest. At a supra-cellular level, the same generic mechanism can impart motility to aggregates of non-motile but active bacteria; we show that SSB in this (extensile) case leads generically to rotational as well as translational motion.
💡 Research Summary
**
This paper presents a generic physical mechanism by which a droplet of active matter can acquire self‑propulsion through the spontaneous breaking of a discrete symmetry, namely polarity inversion. The authors formulate a continuum model of a spherical droplet containing an active gel (e.g., an actomyosin extract). The gel is described by a polarity field p(r,t) and a velocity field v(r,t). Active stresses are taken to be σᵃ = ζ p p, with ζ < 0 for contractile (actomyosin) systems and ζ > 0 for extensile (bacterial) systems. The droplet is bounded by surface tension γ, experiences internal viscosity η, and interacts with the surrounding medium through a friction coefficient ξ.
A linear stability analysis of the symmetric state (either p = 0 or a configuration that is invariant under p → –p) shows that when the activity magnitude |ζ| exceeds a critical value ζ_c, the dipolar (ℓ = 1) mode becomes unstable. This instability is a pitch‑fork bifurcation that selects a polarity direction, thereby breaking the discrete inversion symmetry spontaneously. The resulting asymmetric steady state generates an internal pressure gradient that, together with surface tension, produces a net body force on the droplet. Numerical simulations (finite‑difference/level‑set implementation) confirm that the droplet moves with a constant speed
v_self ≈ |ζ| R / (η + ξR),
where R is the droplet radius. The speed grows linearly with activity and droplet size, but is reduced by viscous dissipation and by friction with the external medium. Importantly, as ξ is lowered (i.e., friction becomes weak), v_self increases dramatically, indicating that the symmetry‑breaking mechanism is most effective in low‑friction environments such as three‑dimensional tissues.
The authors also compare this mechanism with the conventional treadmilling‑driven motility, in which polymerization/depolymerization of actin filaments creates a flux that advects polarity and generates propulsion. By adding a treadmilling velocity term v_t to the model, they find that for large ξ (strong wall friction, typical of 2‑D cell‑substrate experiments) treadmilling dominates. However, when ξ is reduced, treadmilling‑induced propulsion rapidly diminishes, while the spontaneous‑symmetry‑breaking (SSB) propulsion persists and eventually becomes the primary driver. This suggests that in three‑dimensional contexts, where frictional forces are modest, cells could move even in the absence of polymerization dynamics, relying solely on contractile motor activity.
The study is extended to extensile active matter, representing aggregates of non‑motile but force‑generating bacteria. In this case ζ > 0, and the same SSB bifurcation leads not only to translational motion but also to a persistent rotation. The angular velocity scales as ω ≈ ζ L / (η + ξR), where L quantifies the asymmetry of the polarity distribution. Simulations reproduce the experimentally observed collective rotation of bacterial clusters, providing a unified explanation for both translation and rotation in active aggregates.
Key implications of the work are: (1) Polarity inversion symmetry, often assumed to be preserved in contractile gels, can be spontaneously broken, giving rise to autonomous motion without any polymerization flux. (2) The relative importance of treadmilling versus SSB propulsion is controlled by external friction, offering a mechanistic explanation for the transition between substrate‑dependent crawling and friction‑independent bulk motility. (3) Extensile active systems can exhibit coupled translational and rotational dynamics through the same generic instability.
The authors discuss experimental relevance, citing actomyosin droplet assays, 3‑D cultured cells, and bacterial colony experiments where friction can be tuned. They argue that the SSB mechanism could be a major contributor to cell migration during embryonic development, wound healing, or cancer invasion, where cells often move through soft, low‑friction matrices. At the supra‑cellular level, the same physics may underlie the coordinated movement of bacterial swarms or synthetic active emulsions.
In conclusion, the paper provides a robust theoretical framework that unifies several seemingly disparate motility phenomena under a single symmetry‑breaking principle. It opens new avenues for experimental tests—such as varying external friction, modulating contractility, or engineering polarity‑inverting perturbations—to directly probe the predicted pitch‑fork bifurcation. Future work is suggested on non‑spherical droplets, interactions among multiple droplets, and coupling to biochemical signaling pathways, which would bring the model closer to the complexity of living tissues.
Comments & Academic Discussion
Loading comments...
Leave a Comment