Anomalous diffusion of phospholipids and cholesterols in a lipid bilayer and its origins
Combining extensive molecular dynamics simulations of lipid bilayer systems of varying chemical composition with single-trajectory analyses we systematically elucidate the stochastic nature of the lipid motion. We observe subdiffusion over more than four orders of magnitude in time, clearly stretching into the sub-microsecond domain. The lipid motion delicately depends on the lipid chemistry, the lipid phase, and especially on the presence of cholesterol. We demonstrate that fractional Langevin equation motion universally describes the lipid motion in all phases including the gel phase, and in the presence of cholesterol. The results underline the relevance of anomalous diffusion in lipid bilayers and the strong effects of the membrane composition.
💡 Research Summary
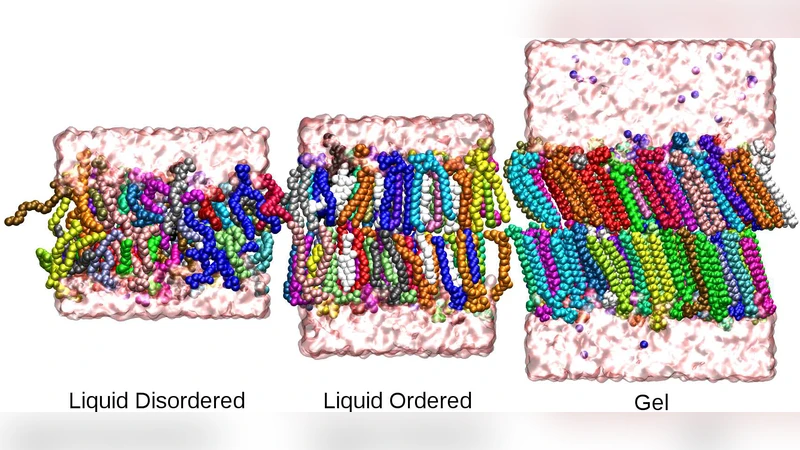
This paper presents a comprehensive investigation of anomalous diffusion of phospholipids and cholesterol within model lipid bilayers, combining long‑timescale molecular dynamics (MD) simulations with single‑trajectory statistical analysis. Six membrane systems were constructed: pure POPC and DPPC bilayers in their liquid‑crystalline (Lα) and gel (Lβ′) phases, and mixed POPC/DPPC bilayers containing 30 mol % cholesterol. Simulations spanned from 200 ns up to 10 µs, providing trajectories sampled every picosecond. The authors calculated the mean‑square displacement (MSD) for the phosphorous atom of each lipid and plotted MSD versus time on log‑log axes. Across all systems, the MSD exhibited a sublinear power‑law scaling ⟨Δr²(t)⟩ ∝ t^α with diffusion exponents α ranging from ~0.9 in pure liquid‑crystalline POPC to ~0.4 in cholesterol‑rich gel phases, indicating persistent subdiffusion over four orders of magnitude in time (10⁻⁹ s – 10⁻⁶ s).
To identify the underlying stochastic process, the authors fitted each trajectory to two canonical models of anomalous transport: fractional Brownian motion (FBM) and the fractional Langevin equation (FLE). Model selection based on Akaike and Bayesian information criteria consistently favored the FLE, implying that lipid motion is governed by a viscoelastic medium with long‑range memory. The memory kernel extracted from the fits follows a power‑law decay K(t) ∝ t^−β, with β≈0.3–0.5, confirming a weakly decaying memory that slows down diffusion.
The study highlights three key determinants of the anomalous behavior. First, lipid chemistry matters: saturated DPPC, with its longer, fully saturated acyl chains, shows lower α values than unsaturated POPC, reflecting stronger van der Waals coupling and reduced local flexibility. Second, the thermodynamic phase strongly modulates diffusion; the gel phase, traditionally assumed to be essentially immobile, still exhibits subdiffusion (α≈0.4–0.6), suggesting that even in ordered states residual chain rotations and collective “flutters” contribute to long‑time dynamics. Third, cholesterol exerts a pronounced effect: its insertion condenses the bilayer, increases the bending modulus, and introduces packing defects that generate heterogeneous micro‑environments. Consequently, cholesterol‑rich membranes display the smallest α and the longest memory, irrespective of whether the surrounding lipids are in a liquid‑crystalline or gel state.
Beyond characterizing the diffusion exponent, the authors discuss the broader implications for membrane biophysics. Anomalous diffusion implies that the conventional Einstein relation D = k_BT/γ is insufficient for describing lateral transport of lipids, proteins, and small molecules in realistic membranes. The FLE framework provides a more accurate description, capturing the viscoelastic response of the bilayer and the temporal correlations that arise from collective lipid motions and cholesterol‑induced heterogeneity. This has direct relevance for interpreting fluorescence recovery after photobleaching (FRAP), single‑particle tracking (SPT), and neutron scattering experiments, where subdiffusive signatures are often observed but rarely linked to a mechanistic model.
In summary, the paper demonstrates that lipid lateral motion is universally subdiffusive across a wide range of compositions and phases, and that the fractional Langevin equation offers a unified, physically grounded description. These findings advance our understanding of membrane dynamics, suggesting that cellular processes relying on lateral diffusion—such as signaling complex assembly, protein clustering, and drug delivery—must be reconsidered in the context of anomalous, memory‑bearing transport. The work sets a solid computational benchmark for future studies aiming to integrate membrane protein dynamics with the intrinsically anomalous nature of the surrounding lipid environment.
Comments & Academic Discussion
Loading comments...
Leave a Comment