Kinetic vs. energetic discrimination in biological copying
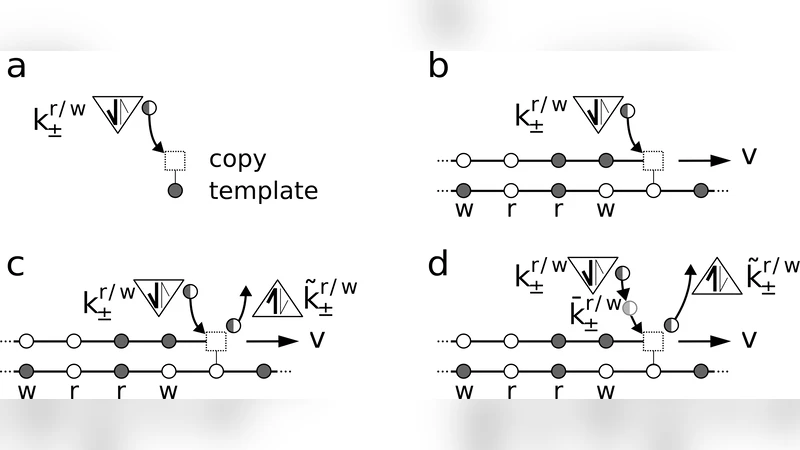
We study stochastic copying schemes in which discrimination between a right and a wrong match is achieved via different kinetic barriers or different binding energies of the two matches. We demonstrate that, in single-step reactions, the two discrimination mechanisms are strictly alternative and can not be mixed to further reduce the error fraction. Close to the lowest error limit, kinetic discrimination results in a diverging copying velocity and dissipation per copied bit. On the opposite, energetic discrimination reaches its lowest error limit in an adiabatic regime where dissipation and velocity vanish. By analyzing experimentally measured kinetic rates of two DNA polymerases, T7 and Pol{\gamma}, we argue that one of them operates in the kinetic and the other in the energetic regime. Finally, we show how the two mechanisms can be combined in copying schemes implementing error correction through a proofreading pathway
💡 Research Summary
The paper investigates how biological copying machines, such as DNA polymerases, achieve high fidelity by discriminating between correct and incorrect substrate matches. Two fundamental discrimination strategies are considered: kinetic discrimination, where the forward transition rates for right and wrong matches differ because of distinct activation barriers, and energetic discrimination, where the forward rates are identical but the final binding free‑energies differ. Using a minimal stochastic model of a single‑step copying reaction, the authors derive exact expressions for the error fraction (ε), the copying velocity (v), and the thermodynamic cost per copied bit (entropy production ΔS).
In the kinetic regime the error rate scales as ε≈exp(−ΔΔG‡/kBT), i.e., it is set by the difference in activation free energies (ΔΔG‡). Because the forward rates can be made arbitrarily large, the velocity can diverge as the error is pushed toward zero, but the energetic cost per bit also diverges. Thus kinetic discrimination can, in principle, achieve arbitrarily low errors only at the price of infinite power dissipation and an unbounded copying speed.
Conversely, in the energetic regime the forward rates for right and wrong incorporations are equal, and the error is governed by the difference in final binding free energies (ΔΔG): ε≈exp(−ΔΔG/kBT). Here the velocity is limited by the common forward rate and vanishes as the error approaches its minimum. The system operates in an adiabatic, quasi‑reversible limit where both the speed and the dissipation per bit go to zero. Hence energetic discrimination yields the lowest possible error with minimal thermodynamic cost, but at the expense of a very slow copying process.
The authors then examine experimental kinetic data for two polymerases: the bacteriophage T7 DNA polymerase and the mitochondrial polymerase Pol γ. T7 exhibits a large disparity between the forward rates for correct and incorrect nucleotides, consistent with a kinetic discrimination mechanism. Pol γ, in contrast, shows similar forward rates but a substantial free‑energy gap between right and wrong bindings, indicating an energetic discrimination strategy. This comparative analysis suggests that nature can employ either mechanism depending on functional requirements.
Finally, the paper extends the analysis to a two‑step copying scheme that incorporates a proofreading pathway. The first step performs the initial nucleotide addition with either kinetic or energetic discrimination, while a second, slower proofreading step can remove misincorporated nucleotides. By coupling the two mechanisms, the overall error can be reduced far below the limits of either single‑step strategy, while the thermodynamic cost and speed are determined by a trade‑off between the discrimination strength of the initial step and the efficiency of proofreading. The authors provide analytical formulas showing how the error, velocity, and dissipation scale with the kinetic and energetic parameters of both steps.
Overall, the study clarifies that kinetic and energetic discrimination are mutually exclusive in a single‑step reaction, each leading to a distinct thermodynamic signature: divergent speed and dissipation for kinetic discrimination versus vanishing speed and dissipation for energetic discrimination. Experimental evidence from T7 and Pol γ supports the coexistence of both strategies in living organisms. Moreover, the work demonstrates that combining the two mechanisms through proofreading offers a powerful route to achieve ultra‑high fidelity with manageable energetic costs, offering valuable design principles for synthetic biology and nanotechnological copying devices.
Comments & Academic Discussion
Loading comments...
Leave a Comment