Physical descriptions of the bacterial nucleoid at large scales, and their biological implications
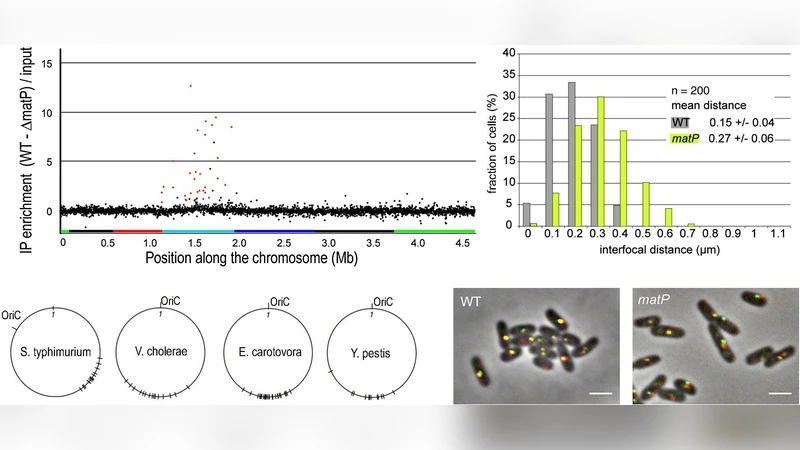
Recent experimental and theoretical approaches have attempted to quantify the physical organization (compaction and geometry) of the bacterial chromosome with its complement of proteins (the nucleoid). The genomic DNA exists in a complex and dynamic protein-rich state, which is highly organised at various length scales. This has implications on modulating (when not enabling) the core biological processes of replication, transcription, segregation. We overview the progress in this area, driven in the last few years by new scientific ideas and new interdisciplinary experimental techniques, ranging from high space- and time-resolution microscopy to high-throughput genomics employing sequencing to map different aspects of the nucleoid-related interactome. The aim of this review is to present the wide spectrum of experimental and theoretical findings coherently, from a physics viewpoint. We also discuss some attempts of interpretation that unify different results, highlighting the role that statistical and soft condensed matter physics, and in particular classic and more modern tools from the theory of polymers, plays in describing this system of fundamental biological importance, and pointing to possible directions for future investigation.
💡 Research Summary
This review synthesizes recent experimental and theoretical advances that aim to quantify the physical organization of the bacterial chromosome together with its associated proteins, collectively known as the nucleoid. The authors begin by emphasizing that the nucleoid is not a static, homogeneous mass of DNA but a highly dynamic, protein‑rich polymer network that exhibits organization across multiple length scales—from nanometer‑scale DNA fibers to micrometer‑scale domains. Cutting‑edge microscopy techniques, such as super‑resolution PALM/STORM, lattice light‑sheet imaging, and high‑speed single‑particle tracking, have revealed that bacterial DNA adopts a series of compacted loops and supercoiled plectonemes that are interspersed with protein‑dense “droplets.” These droplets, enriched in nucleoid‑associated proteins (NAPs) such as H‑NS, Fis, and HU, behave like liquid‑like condensates, creating regions of high local viscosity and reduced DNA mobility.
Complementary to imaging, high‑throughput genomic methods—including Hi‑C, Micro‑C, and ChIP‑seq—have mapped DNA‑DNA contact frequencies and protein binding sites genome‑wide. The contact maps display a hierarchical pattern: short‑range contacts are dominated by NAP‑mediated loops of 10–100 kb, while longer‑range interactions reflect the global compaction imposed by structural maintenance of chromosomes (SMC) complexes. Notably, the authors point out that the distribution of loop sizes follows a non‑Gaussian, scale‑free law, suggesting that the nucleoid operates near a critical point where small perturbations can reorganize large portions of the genome.
From a theoretical perspective, the review integrates classic polymer physics (self‑avoiding walks, Flory theory) with modern concepts such as active loop extrusion, topological constraints, and phase separation. The authors propose an “active loop‑extrusion plus phase‑separation” framework: SMC complexes act as motor proteins that extrude loops, thereby generating tension and reducing supercoiling, while NAPs act as cross‑linkers that promote microphase separation into dense protein‑DNA droplets and dilute DNA‑rich regions. Monte‑Carlo and Langevin dynamics simulations based on this framework successfully reproduce experimental observables, including the scaling of nucleoid radius with genome length, the emergence of heterogeneous diffusion coefficients, and the rapid re‑organization observed during replication fork progression.
Biological implications are explored in depth. During DNA replication, the advancing fork creates excess twist; topoisomerases and SMC complexes cooperate to dissipate this torsional stress, preventing fork stalling. Transcriptionally active loci are preferentially located at the periphery of dense droplets, where the local environment is more fluid, facilitating RNA polymerase access. Conversely, H‑NS‑rich regions form repressive condensates that physically impede transcription machinery, providing a mechanistic link between nucleoid architecture and gene regulation. In cell division, the nucleoid is compacted by SMC‑driven loop extrusion, ensuring equal partitioning of genetic material into daughter cells; this compaction follows a scaling law that ties nucleoid volume to cell size, allowing bacteria to adapt nucleoid organization to growth conditions.
Finally, the review outlines future directions. The authors advocate for integrated multimodal approaches that combine real‑time 3D super‑resolution imaging with single‑molecule force spectroscopy to capture nucleoid dynamics on sub‑second timescales. They also highlight the need for systems‑level modeling that incorporates transcriptomics, proteomics, and epigenetic modifications to predict how environmental stresses reshape nucleoid architecture. By bridging experimental data with polymer‑physics‑based models, the field moves toward a unified, quantitative description of the bacterial nucleoid as a soft‑condensed‑matter system that orchestrates essential cellular processes.
Comments & Academic Discussion
Loading comments...
Leave a Comment