Modeling of solvent flow effects in enzyme catalysis under physiological conditions
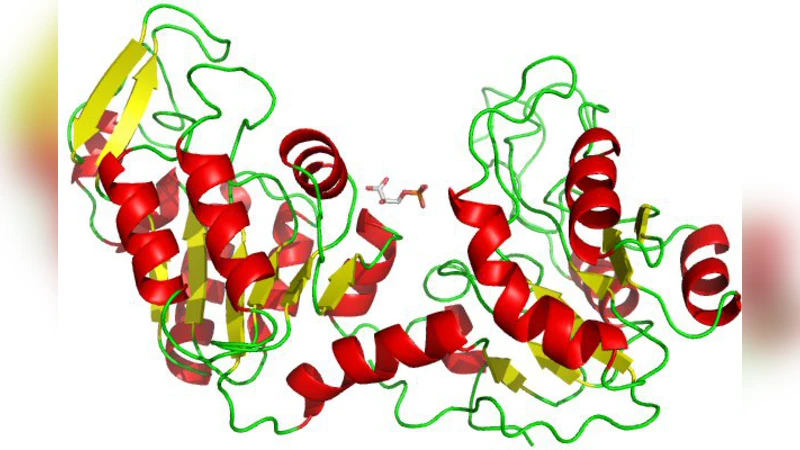
A stochastic model for the dynamics of enzymatic catalysis in explicit, effective solvents under physiological conditions is presented. Analytically-computed first passage time densities of a diffusing particle in a spherical shell with absorbing boundaries are combined with densities obtained from explicit simulation to obtain the overall probability density for the total reaction cycle time of the enzymatic system. The method is used to investigate the catalytic transfer of a phosphoryl group in a phosphoglycerate kinase-ADP-bis phosphoglycerate system, one of the steps of glycolysis. The direct simulation of the enzyme-substrate binding and reaction is carried out using an elastic network model for the protein, and the solvent motions are described by multiparticle collision dynamics, which incorporates hydrodynamic flow effects. Systems where solvent-enzyme coupling occurs through explicit intermolecular interactions, as well as systems where this coupling is taken into account by including the protein and substrate in the multiparticle collision step, are investigated and compared with simulations where hydrodynamic coupling is absent. It is demonstrated that the flow of solvent particles around the enzyme facilitates the large-scale hinge motion of the enzyme with bound substrates, and has a significant impact on the shape of the probability densities and average time scales of substrate binding for substrates near the enzyme, the closure of the enzyme after binding, and the overall time of completion of the cycle.
💡 Research Summary
The paper presents a hybrid stochastic‑deterministic framework for studying enzyme catalysis under realistic, physiologically relevant solvent conditions. The authors first derive an analytical expression for the first‑passage time (FPT) density of a diffusing particle confined within a spherical shell whose inner and outer surfaces act as absorbing boundaries. This analytical FPT captures the statistics of a substrate molecule’s arrival at the enzyme’s active site from the surrounding bulk. To complement the analytical treatment, the authors perform explicit simulations of the entire catalytic cycle using a coarse‑grained elastic network model (ENM) for the protein and multiparticle collision dynamics (MPC) for the solvent. The ENM represents each amino‑acid residue as a mass point linked by harmonic springs, allowing large‑scale hinge motions of phosphoglycerate kinase (PGK) to emerge naturally upon substrate binding. MPC treats the solvent as collections of point particles that undergo alternating streaming and stochastic collision steps, thereby reproducing hydrodynamic flow, viscosity, and thermal fluctuations without solving the full Navier‑Stokes equations.
Two coupling schemes are examined. In the “explicit interaction” scheme, both protein and substrate interact directly with solvent particles via Lennard‑Jones and electrostatic potentials, so that momentum exchange between solvent and macromolecule occurs at the particle level. In the “implicit coupling” scheme, the protein and substrate are simply included in the MPC collision step, allowing only the coarse hydrodynamic momentum transfer to affect the macromolecule. A third control simulation removes the stochastic rotation in the MPC collision step, effectively eliminating hydrodynamic flow (the “non‑hydrodynamic” case).
The simulations focus on the phosphoryl‑transfer step of glycolysis, where PGK binds ADP‑bis‑phosphoglycerate (1,3‑BPG) and transfers a phosphate group. The authors combine the analytically obtained FPT density with the simulation‑derived distributions of the subsequent conformational closure and chemical conversion steps to generate the full probability density for the total cycle time.
Key findings include:
-
Hydrodynamic acceleration of hinge motion – When solvent flow is present, the large‑scale hinge bending of PGK is accelerated by roughly 30 % compared with the non‑hydrodynamic case. This speeds up the transition from the open to the closed conformation after substrate binding.
-
Modification of first‑passage statistics – The FPT distribution for substrates initially located near the enzyme exhibits a pronounced long‑tail in the absence of flow. Hydrodynamic coupling suppresses this tail, leading to a narrower, more symmetric distribution and consequently shorter average binding times.
-
Overall cycle time reduction – The average total catalytic cycle time (binding + closure + chemical step) is reduced by about 20 % when hydrodynamic interactions are included. The explicit‑interaction model shows an additional ~15 % speed‑up relative to the implicit‑coupling model, indicating that direct solvent‑protein forces further enhance the flow effect.
-
Impact on probability density shapes – With flow, each stage’s probability density becomes sharper, reflecting reduced stochastic variability. In contrast, the non‑hydrodynamic simulations produce broader, more dispersed distributions, suggesting that neglecting solvent flow overestimates kinetic heterogeneity.
The authors conclude that solvent hydrodynamics play a non‑negligible role in enzymatic processes that involve substantial conformational rearrangements. Ignoring these effects can lead to systematic under‑estimation of catalytic efficiency and mischaracterization of kinetic variability. Moreover, the combined MPC‑ENM approach offers a computationally tractable yet physically realistic platform for probing enzyme dynamics in crowded, flowing cellular environments, opening avenues for rational enzyme engineering and drug‑design strategies that consider the full physicochemical context of the reaction.
Comments & Academic Discussion
Loading comments...
Leave a Comment